Advertisements
Advertisements
Question
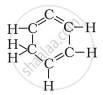
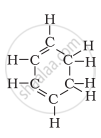
Structural formula of benzene is
Options
Advertisements
Solution

The formula of benzene is C6H6. It has three alternate pi bonds in the ring of carbon atoms.
APPEARS IN
RELATED QUESTIONS
Fill in the blank.
The name ‘carbon’ is derived from the Latin word...........
How many carbon atoms are there in Buckminster fullerenes?
Match the colunm
| column A | column B |
| 1. A product of incomplete burning | a. Hopcolite |
| 2. Nature of carben monoxide | b. Combustible gas |
| 3. A compound formed by the combination of haemoglobin and carbon monoxide | c. carboxy-haemoglobin |
| 4. A mixture of metallic oxides | d. Carbon monoxide |
| 5. Carbon monoxide | e. Highly poisonous |
FILL IN THE BLANK
Carbon atom has ................... electrons in its valence shell.
FILL IN THE BLANK
..................... is used as a pigment and colouring matter in ink and shoe polish.
What are hydrocarbons ? Name the two different forms of hydrocarbons.
What is the name of the famous diamond that was found in Wajrakarur in Andhra Pradesh ?
Write three chemical properties of carbon.
______ is the essential element in all the organic compounds.
Answer the following question.
Why are methane compounds have poor conductors of electricity?
Answer the following question.
What happens when methane compound burns in oxygen?
How would you prove experimentally that carbon dioxide does not support combustion.
Starting from carbon dioxide how would you obtain a weak acid.
[Give balanced equations for the same]
Give reason for the following:
Both CO2 & SO2 turn lime water milky. Moist potassium permanganate paper, helps in distinguishing the two gases.
Higher Order Thinking Skill.
Why do carbon exist mostly in combined state?
Vinegar is a solution of
The main component of water gas is ______.
A large family of fullerene exists, starting at ______ and ______ reaching up to.
Answer the following by rearranging the jumbled letters :
In this molecule, carbon is attached to four hydrõgen atoms.
emathen.
Why is carbon monoxide harmful to health? (or) What happens when we breathe Carbon monoxide?