Advertisements
Advertisements
Question
State your observation for the following case:
Paper soaked in potassium permanganate solution is introduced into a gas jar of sulphur dioxide.
Advertisements
Solution
The bleaching effect of sulphur dioxide causes the pink tint of potassium permanganate paper to turn colourless.
RELATED QUESTIONS
Write balanced chemical equations to show : The oxidizing action of conc. Sulphuric acid on carbon
Write balanced chemical equations to show how SO3 is converted to Sulphuric acid in the contact process.
The acid which is used in the preparation of a volatile acid
Give balanced chemical equations for the action of sulphuric acid on the following:
Potassium hydrogen carbonate.
Give reason for the following:
H2SO4 cannot be prepared by heating sodium sulphate with conc. HCl or HNO3
Write balanced equation for the reaction of dilute sulphuric acid with the following:
Zinc hydroxide
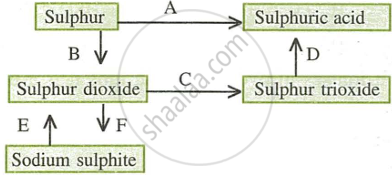
- Name the catalyst which helps in the conversion of sulphur dioxide to sulphur trioxide in step C.
- In the contact process for the manufacture of sulphuric acid, sulphur trioxide is not converted to sulphuric acid by reacting it with water. Instead a two-step procedure is used. Write the equations for the two steps involved in D.
- What type of substance will liberate sulphur dioxide from sodium sulphite in step E?
- Write the equation for the reaction by which sulphur dioxide is converted to sodium sulphite in step F.
Write the equation of the following reaction :
Concentrated sulphuric acid is poured over sugar
Write the observation for the following:
Decomposition of bicarbonates by dil. H2SO4.
\[\ce{2NaHCO3 + H2SO4 -> Na2SO4 + 2H2O + 2CO2}\]
Complete the table given below:
| Name of the process | Reactants | Acid product formed |
| (a) ______ | Nitrogen dioxide + water + oxygen | (b) ______ |
| (c) ______ | Oleum + water | Sulphuric acid |
