Advertisements
Advertisements
Question
State the importance of the solubility of CO2 and O2 in water.
Advertisements
Solution
- Fish extracts the oxygen from the water and expels water through the gills. Fish can survive in water only through the dissolved oxygen present in water.
- Aquatic plants make use of dissolved carbon dioxide for photosynthesis.
- Carbon dioxide dissolved in water reacts with limestone to form calcium bicarbonate.
- Marine organisms such as snails, oysters, etc., extract calcium carbonate from calcium bicarbonate to build their shells.
APPEARS IN
RELATED QUESTIONS
Water has ...................... density and .................. volume at 4°C.
The solubility of a gas in water.............. with rise in temperature and.......................with rise in pressure.
The water in a lake is called ground water.
State any two sources of water pollution.
Pour water from the ice tray into a kettle. Ask an elder at your home to heat the kettle and observe the mouth of the kettle when the water starts boiling.
What do you see?
Look at the map of Tamilnadu showing annual rainfall and answer the questions given below

- Identify the districts that get only low annual rainfall in Tamilnadu.
- Identify the districts that get a medium annual rainfall in Tamilnadu.
- State the districts that enjoy high annual rainfall in Tamilnadu.
Total earth’s surface covered by water is
Match Column I with Column II
| Column I | Column II | ||
| (a) | Ground water | (i) | Solid form of water |
| (b) | Bawri | (ii) | Wastage of water |
| (c) | Snow | (iii) | Water management |
| (d) | Drip irrigation | (iv) | Small water |
| (e) | Leaking taps | (v) | Water table |
Complete the following chart by writing appropriate words in the boxes marked (a) to (e).
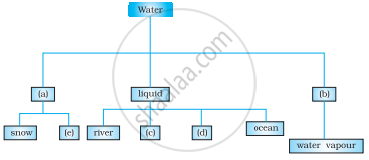
Water in our tap comes from a ______.
