Advertisements
Advertisements
Question
State reasons for the following:
Aliphatic amines are stronger bases than aromatic amines.
Give Reasons
Advertisements
Solution
Aliphatic amines have an alkyl group that raises electron density on the N atom by (+1) induction. This makes the –NH2 group more basic and attracts a proton.
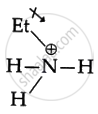
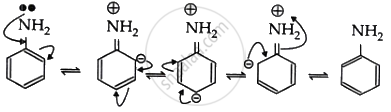
As a result, the lone pair of electrons in an aromatic amine is involved in the benzene ring’s resonance and cannot display basic character. So, aliphatic amines are more basic than aromatic amines.
shaalaa.com
Is there an error in this question or solution?
2019-2020 (March) Official
