Advertisements
Advertisements
Question
Select from the list given (a to e) one substances in each case which matches the description given in parts (i) to (v). (Note : Each substance is used only one in the answer)
(a) Nitroso Iron (II) Sulphate
(b) Iron (III) chloride
(c) Chromium sulphate
(d) Lead (II) chloride
(e) Sodium chloride
(i) A compound which is deliquescent
(ii) A compound which is insoluble in cold water, but soluble in hot water
(iii) The compound responsible for the brown ring during the brown ring test of nitrate iron
(iv) A compound whose aqueous solution is neutral in nature
(v) The compound which is responsible for the green colouration when sulphur dioxide is passed through acidified potassium dichromate solution
Advertisements
Solution
(i) Iron (III) chloride
(ii) Lead (II) chloride
(iii) Nitroso iron (II) sulphate
(iv) Sodium chloride
(v) Chromium sulphate
APPEARS IN
RELATED QUESTIONS
State your observation when dilute hydrochloric acid is added to a lead nitrate solution and the mixture is heated.
State your observations when Dilute Hydrochloric acid is added to Copper carbonate
State your observations when Dilute Hydrochloric acid is added to Sodium thiosulphate.
Select from the list the gas that matches the description given in case
[ammonia, ethane, hydrogen chloride, hydrogen sulfide, ethyne]
This gas has a characteristic rotten egg smell.
State your observation When moist starch iodide paper is introduced into chlorine gas.
What would happen if
Calcium oxide is used to dry hydrogen chloride.
A solution of hydrogen chloride in water is prepared. The following substances are added to separate portions of the solution:
| S. No. | Substances added | Gas evolved | Odour |
| 1. | Calcium carbonate | _________ | _________ |
| 2. | Magnesium ribbon | _________ | _________ |
| 3. | Manganese (IV) oxide with heating | _________ | _________ |
| 4. | Sodium sulphide | _________ | _________ |
Complete the table by writing the gas evolved in each case and its odour.
Write a fully balanced of the following case :
Red lead is warmed with concentrated hydrochloric acid.
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D, and E.
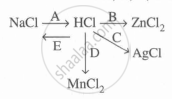
State which of the two - a solution of HCl in water or in toluene is an electrolyte. Explain.
