Advertisements
Advertisements
Question
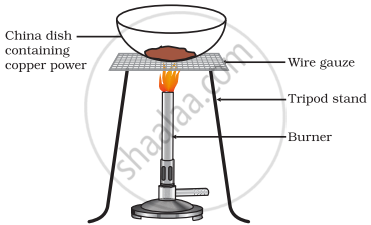
Seema took a small amount of copper powder in a china dish and heated it. What changes will she observe on heating? When H2 gas is passed over this heated substance, what visible changes will be seen in it? Give the chemical equations of the reactions involved, along with the names and colours of the products in each case.
Advertisements
Solution
On heating the copper powder, the reddish-brown copper powder turns black due to the formation of copper(II) oxide (CuO). This is an oxidation reaction.
\[\ce{2Cu + O2 ->[heat] 2CuO}\]
When H2 gas is passed over the heated substance, the black copper oxide turns back to brown copper. This is a reduction reaction (CuO is reduced, H2 is oxidised).
\[\ce{CuO + H2 -> Cu + H2O}\]
In this process, oxidation and reduction both reactions take place, forming a redox reaction.
