Advertisements
Advertisements
Question
Read the following passage and answer the questions that follow:
|
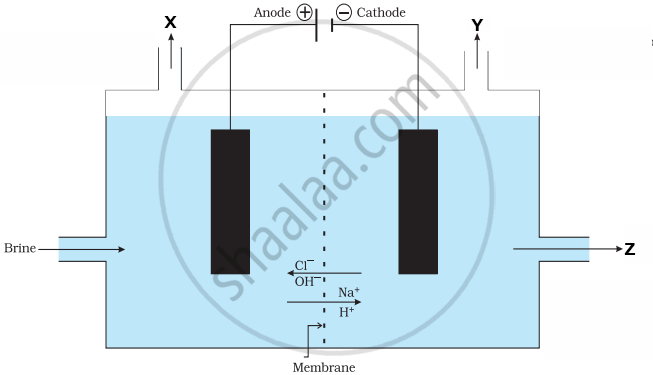
In the given diagram, when electricity is passed through an aqueous solution of a common salt, a substance ‘Z’ is produced, along with the evolution of gases ‘X’ and ‘Y’. When a burning matchstick is brought near the gas ‘Y’, it burns with a pop sound, whereas ‘X’ is used for disinfecting drinking water. When ‘X’ is passed through slaked lime, an insoluble substance ‘A’ is produced.
|
(a) Write the names of the gases ‘X’ and ‘Y’. [1]
(b) Write the balanced chemical equation for the formation of substance ‘Z’. [1]
(c) (i) What happens: [2]
- If a drop of red litmus solution is added to the aqueous solution of substance ‘Z’?
- If CO2 gas is passed through ammoniacal solution of aqueous NaCl?
OR
(c) (ii) Identify the substance ‘A’ and write the balanced chemical equation of the reaction which takes place in its preparation. [2]
Advertisements
Solution
(a) X: Chlorine gas (Cl2)
Y: Hydrogen gas (H2)
(b) \[\ce{2NaCl_{(aq)} + 2H2O_{(l)} ->[electric current] 2NaOH_{(aq)} + H2_{(g)} + Cl2_{(g)}}\]
(c) (i) (I) If a drop of red litmus solution is added to the aqueous solution of substance ‘Z’, it turns blue, showing that NaOH is basic.
(II) If CO2 gas is passed through an ammoniacal solution of aqueous NaCl, Sodium hydrogen carbonate (NaHCO3) precipitate is formed (Solvay process).
\[\ce{NaCl + NH3 + H2O + CO2 -> NaHCO3\downarrow + NH4Cl}\]
OR
(c) (ii) Substance ‘A’ is bleaching powder (CaOCl2).
\[\ce{Ca(OH)2 + Cl2 -> CaOCl2 + H2O}\]
Formed when chlorine gas is passed through slaked lime.