Advertisements
Advertisements
Question
Obtain Bohr’s quantisation condition for angular momentum of electron orbiting in nth orbit in hydrogen atom on the basis of the wave picture of an electron using de Broglie hypothesis.
Advertisements
Solution 1
`n lambda = 2 pi r`
But ` lambda = n/(mv) `( By De Broglie)
`(n lambda) /(mv) = 2 pi r`
` therefore (lambda n) /(2 pi ) = mvr ` (mvr = L)
` therefore L = (lambda n)/(2 pi)`
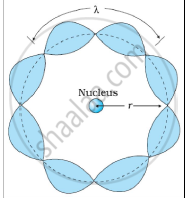
Solution 2
Bohr's second postulate states that the angular momentum of an electron has only those values that are integral multiples of `"h"/(2pi)` He thought that the motion of electrons within an atom is associated with the standing wave along the orbit as shown.
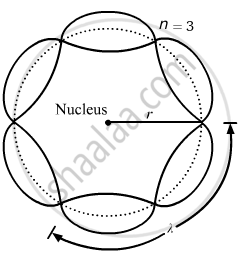
About standing waves in stretched strings, we know that only those waves survive for which the distances traveled in the round trip between the ends are integral multiples of the wavelength. Similarly, for an electron moving in the nth orbit of radius rn, the distance traveled in one trip is 2πrn, which should be an integral multiple of the wavelength.
2πrn = nλ (where n= 1, 2, 3, 4 etc)
By de-Broglie hypothesis, we have:
`lambda = "h"/"p" = "h"/("mv"_"n")`
Substituting the value of λ in the above expression, we get:
`2pi"r"_"n" = "n" "h"/"mv"_"n"`
⇒ `"mv"_"n""r"_"n" = "n" "h"/(2pi)`
Angular momentum `= "L" = "n" "h"/(2pi)`
RELATED QUESTIONS
Find the frequency of revolution of an electron in Bohr’s 2nd orbit; if the radius and speed of electron in that orbit is 2.14 × 10-10 m and 1.09 × 106 m/s respectively. [π= 3.142]
What is the maximum number of emission lines when the excited electron of an H atom in n = 6 drops to the ground state?
Using Bohr's postulates, derive the expression for the orbital period of the electron moving in the nth orbit of hydrogen atom ?
Which of the following parameters are the same for all hydrogen-like atoms and ions in their ground states?
When a photon stimulates the emission of another photon, the two photons have
(a) same energy
(b) same direction
(c) same phase
(d) same wavelength
A filter transmits only the radiation of wavelength greater than 440 nm. Radiation from a hydrogen-discharge tube goes through such a filter and is incident on a metal of work function 2.0 eV. Find the stopping potential which can stop the photoelectrons.
Calculate the de-Broglie wavelength associated with the electron revolving in the first excited state of the hydrogen atom. The ground state energy of the hydrogen atom is −13.6 eV.
Ratio of longest to shortest wavelength in Balmer series is ______.
An ionised H-molecule consists of an electron and two protons. The protons are separated by a small distance of the order of angstrom. In the ground state ______.
- the electron would not move in circular orbits.
- the energy would be (2)4 times that of a H-atom.
- the electrons, orbit would go around the protons.
- the molecule will soon decay in a proton and a H-atom.
In Bohr's theory of hydrogen atom, the electron jumps from higher orbit n to lower orbit p. The wavelength will be minimum for the transition ______.
