Advertisements
Advertisements
Question
Name the technique that you use if only the solid component of a solution is required.
Advertisements
Solution
Evaporation.
APPEARS IN
RELATED QUESTIONS
Tick the most appropriate answer
Solutions are
- heterogeneous mixtures.
- compounds
- homogeneous mixtures.
- elements
Give the symbols of: Carbon, calcium, copper, chlorine, cobalt, argon.
Give two examples of Noble gases.
Give reason: A frying pan is made up of steel but its handle is made up of wood.
Indicate whether the following statement is true or false:
Molecules combine to form atoms
Tick (√) the correct alternative from the choice given for the following statement.
All pure substances have
The important physical properties of substances are colour, odour, nature, density & solubility in water.
Name – a gas with a pungent, choking odour which is lighter than air
Complete the statement given below by filling in the blanks with the correct words.
Atom contains ________ , with positively charged _____.
Select the correct answer from the choice given below.
An impure substance
Write the names of elements from the following symbol:
Si
Write the names of elements from the following symbol:
Ir
‘The modern periodic table consists of elements arranged according to their increasing atomic numbers’. With reference to elements with atomic numbers 1 to 20 only in the periodic table – differentiate them into – metallic elements, metalloids, non-metals & noble gases.
State why brass is considered as a homogenous mixture while a mixture of iron & sulphur – heterogenous.
Select the correct answer from A, B, C, D & E for the statement given below :
A diatomic molecule.
The diagram represents fractional distillation for the separation of mixtures. Answer the following :
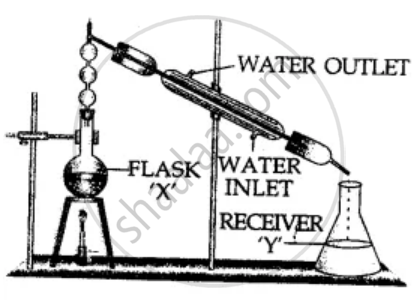
- Can two immiscible liquids be separated by this process?
- Separation of liquids by this process is based on which physical property?
- If methyl alcohol & water are to be separated, which liquid would remain in flask ‘X’ after condensation.
- Give a reason for the above answer.
- State the purpose of the fractionating column in the apparatus.
State true of false. If false, give the correct statement.
Two different elements may have similar atoms.
What are elements? What are they made of?
Name the element which is used for making computer chips.
Higher-Order Thinking questions:
Why is hydrogen considered an element?
