Advertisements
Advertisements
Question
Name the following:
Salt obtained by heating sodium chloride with concentrated sulphuric acid below 200 degree celcius.
Advertisements
Solution
Salt obtained by heating sodium chloride with concentrated sulphuric acid below 200 degree Celsius is sodium hydrogen sulphate.
\[\ce{\underset{\text{Sodium chloride}}{NaCl} +\underset{\text{Sulphuric Acid(Conc.)}}{H2SO4} ->[<200°C] \underset{\text{Sodium hydrogen sulphate}}{NaHSO4} + \underset{\text{Hydrogen gas}}{HCl}}\]
APPEARS IN
RELATED QUESTIONS
State one relevant observation for given reactions:
Action of dilute Hydrochloric acid on iron (II) sulfide.
State your observations when Dilute Hydrochloric acid is added to Sodium thiosulphate.
Name an element which reacts with hydrogen to form a compound which is strongly acidic in water.
MnO2, PbO2 and red lead react with conc. HCl acid to liberate Cl2.
What is the common property being shown by these metal oxides?
Name the following:
Five oxidizing agents that liberated chlorine from concentrated hydrochloric acid.
Fill in the blank:
On addition of silver nitrate to hydrochloric acid ___________ precipitate is formed which is soluble in ____________
Write a fully balanced of the following case :
Magnesium metal is treated with dilute hydrochloric acid.
Write the balanced equation for the reaction of dilute hydrochloric acid with the following:
Iron (II) sulphide
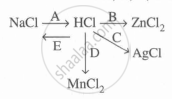
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D, and E.
State which of the two - a solution of HCl in water or in toluene is an electrolyte. Explain.
