Advertisements
Advertisements
Question
Mention two possible ways by which ammonia produced is removed from unchanged gases.
Advertisements
Solution
The gases leaving the catalyst chamber are cooled by passing through
condensing pipes where ammonia gets liquefied and is collected in
receiver.
Key: Haber's Process
APPEARS IN
RELATED QUESTIONS
Ammonium salts decompose on heating. What other property do ammonium salts have in common?
Name two gases which give dense white fumes with ammonia.
How will you demonstrate the solubility of ammonia in water? Explain.
State your observation for the following cases
(i) Ammonia gas is burnt in an atomosphere of oxygen in the absence of a catalyst
(ii) Glass rod dipped in ammonium hydroxide is brought near the mouth of the concentrated hydrochloric acid bottle
Identify the substance underlined, in the following case:
A solid formed by reaction of two gaes, one of which is acidic and the other basic in nature.
Given one test can be used to detect the presence of the ion produced
The diagram shows an experimental set up for the laboratory preparation of a pungent smelling gas. The gas is alkaline in nature.
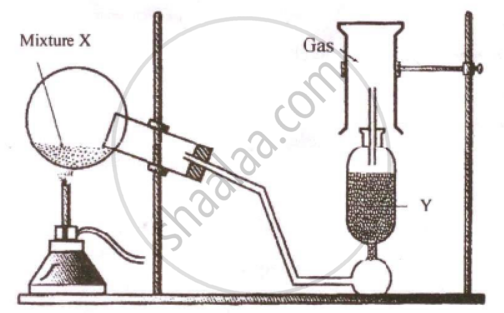
- Nature the gas collected in the jar.
- Write the balance equation for the above preparation.
- How is the gas being collected?
- Name the drying agent used.
- How will you find that the jar is full of gas?
What do you observe when Ammonium solution is added to ferric chloride solution.
What do you observe when Ammonia solution is added drop by drop and then in excess to aqueous copper sulphate solution.
