Advertisements
Advertisements
Question
Match the structures of compounds given in Column I with the classes of compounds given in Column II.
| Column I | Column II | |
| (i) | \[\begin{array}{cc} \ce{CH3 - CH - CH3}\\ |\phantom{..}\\ \ce{X}\phantom{..} \end{array}\] |
(a) Aryl halide |
| (ii) | \[\ce{CH2 = CH - CH2 - X}\] | (b) Alkyl halide |
| (iii) | 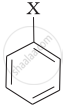 |
(c) Vinyl halide |
| (iv) | \[\ce{CH2 = CH - X}\] | (d) Allyl halide |
Advertisements
Solution
| Column I | Column II | |
| (i) | \[\begin{array}{cc} \ce{CH3 - CH - CH3}\\ |\phantom{..}\\ \ce{X}\phantom{..} \end{array}\] |
(b) Alkyl halide |
| (ii) | \[\ce{CH2 = CH - CH2 - X}\] | (d) Allyl halide |
| (iii) |  |
(a) Aryl halide |
| (iv) | \[\ce{CH2 = CH - X}\] | (c) Vinyl halide |
Explanation:
(i) In alkyl halide, the halogen atom is bonded to sp3 hybridised carbon atom, which may be further bonded to one, two or three alkyl groups, i.e., \[\begin{array}{cc}
\ce{CH3 - CH - CH3}\\
|\phantom{..}\\
\ce{X}\phantom{..}
\end{array}\]
(ii) Allyl halides are the compounds in which the halogen atom is bonded to sp3 hybridised carbon atom next to the carbon-carbon double bond, i.e., \[\ce{CH2 = CH - CH2 - X}\]
(iii) Aryl halides are the compounds in which the halogen atom is bonded to the sp2 hybridised carbon atom of an aromatic ring, i.e., \[\ce{C6H5X}\]
(iv) Vinyl halides are the compounds in which the halogen atom is bonded to an sp2 hybridised carbon atom of a carbon-carbon double bond, i.e., \[\ce{CH2 = CH-X}\]
APPEARS IN
RELATED QUESTIONS
Draw the structure of the major monohalo product in the following reaction:

Draw the structure of the major monohalo product in the following reaction:

Draw the structure of the major monohalo product in the following reaction:

In which of the following molecules carbon atom marked with asterisk (*) is asymmetric?
(a) \[\begin{array}{cc}
\ce{H}\\
|\\
\phantom{.}\ce{C^*}\phantom{}\\
\phantom{}/\phantom{..}|\phantom{..}\backslash\phantom{}\\
\phantom{.}\ce{I}\phantom{...}\ce{Br}\phantom{..}\ce{Cl}
\end{array}\]
(b) \[\begin{array}{cc}
\ce{D}\\
|\\
\phantom{.}\ce{C^*}\phantom{}\\
\phantom{}/\phantom{..}|\phantom{..}\backslash\phantom{}\\
\phantom{.}\ce{I}\phantom{...}\ce{Br}\phantom{..}\ce{Cl}
\end{array}\]
(c) \[\begin{array}{cc}
\ce{H}\\
|\\
\phantom{.}\ce{C^*}\phantom{}\\
\phantom{}/\phantom{..}|\phantom{..}\backslash\phantom{}\\
\phantom{..}\ce{OH}\phantom{..}\ce{C2H5}\phantom{}\ce{CH3}
\end{array}\]
(d) \[\begin{array}{cc}
\ce{H}\\
|\\
\phantom{.}\ce{C^*}\phantom{}\\
\phantom{}/\phantom{..}|\phantom{..}\backslash\phantom{}\\
\phantom{..}\ce{H}\phantom{...}\ce{C2H5}\phantom{}\ce{CH3}
\end{array}\]
The position of \[\ce{-Br}\] in the compound in \[\ce{CH3CH = CH(Br)(CH3)2}\] can be classified as ______.
Which of the products will be major product in the reaction given below? Explain.
\[\ce{CH3CH = CH2 + HI -> \underset{(A)}{CH3CH2CH2I} + \underset{(B)}{CH3CHICH3}}\]
Which alkyl halide has maximum density
Why are haloalkanes more reactive towards nucleophilic substitution reactions than haloarenes and vinylic halides?
Name the following halide according to IUPAC system and classify it as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halide:
\[\ce{CH3C(C2H5)2CH2Br}\]
Name the following halide according to the IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl, or aryl halide:
\[\ce{CH3CH2C(CH3)2CH2I}\]
Name the following halides according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3CH2 C(CH3)2CH2I}\]
Name the following halide according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3C(C2H5)2CH2Br}\]
Name the following halide according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3CH2C(CH3)2CH2I}\]
Name the following halides according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3CH2C(CH3)2CH2I}\]
Give the IUPAC name of the compound.
\[\ce{CICH2 C ≡ CCH2 Br}\]
Name the following halide according to IUPAC system and classify as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3C(C2H5)2CH2Br}\]
Name the following halides according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3C(Cl)(C2H5)CH2CH3}\]
Which of the following is an example of vicdihalide?
Ethylidene chloride is a/an ______.
