Advertisements
Advertisements
Question
List the properties of ammonia that make it
(i) A good refrigerant
(ii) A cleaning agent
(iii) As a source of hydrogen
Advertisements
Solution
(i) Liquid ammonia is used as refrigerant as:
(a) It is highly volatile
(b) It has high specific latent heat of vaporization. 1 mole (17g) of liquid ammonia vaporises by absorbing 5.7 kcals of heat from the surroundings, which is there by cooled.
(c) It easily liquefies under pressure at room temperature.
(ii) Ammonia emulsifies fats and grease. Thus it is used to clean oils, fats and body grease etc. from clothes.
(iii) Liquid hydrogen is dangerous to transport as it is highly combustible. Thus, hydrogen is converted to liquid ammonia and transported in cylinders. Later it is catalytically converted to hydrogen.
Key: Uses of ammonia
APPEARS IN
RELATED QUESTIONS
What property of ammonia is demonstrated by the fountain experiment?
Name two gases which give dense white fumes with ammonia.
Mention two possible ways by which ammonia produced is removed from unchanged gases.
Choose the correct word or phrase from the bracket to complete the following sentences.
Heating solution of ammonium chloride with sodium nitrite produces ______
Name the other ion formed when ammonia dissolves in water
The diagram shows an experimental set up for the laboratory preparation of a pungent smelling gas. The gas is alkaline in nature.
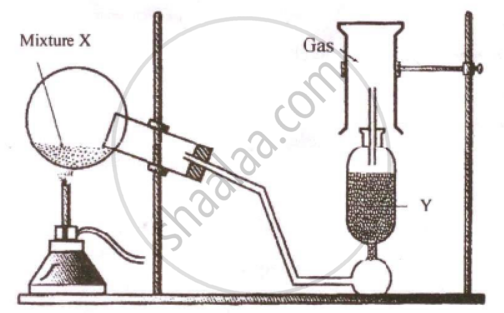
- Nature the gas collected in the jar.
- Write the balance equation for the above preparation.
- How is the gas being collected?
- Name the drying agent used.
- How will you find that the jar is full of gas?
Choose the correct answer from the options given below :
Ammonia is produced when ammonium chloride is heated with
- Of the two gases, ammonia and hydrogen chloride, which is more dense? Name the method of collection of this gas.
- Give one example of a reaction between the above two gases which produce a solid compound.
- Write the balanced equation for a reaction in which ammonia is oxidized by:
- a metal oxide,
- a gas which is not oxygen.
