Advertisements
Advertisements
Question
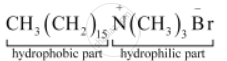
Label the hydrophilic and hydrophobic parts in the following compounds.

Advertisements
Solution
RELATED QUESTIONS
Explain the following term with a suitable example:
Cationic detergents
Explain the following terms with suitable examples - Non-ionic detergents
What are biodegradable and non-biodegradable detergents? Give one example of each.
If water contains dissolved calcium hydrogen carbonate, out of soaps and synthetic detergents which one will you use for cleaning clothes?
Label the hydrophilic and hydrophobic parts in the following compounds.
CH3(CH2)10CH2OSO3 –Na+
What are anionic detergents? Give an example ?
Polyethyleneglycols are used in the preparation of which type of detergents?
Draw the diagram showing micelle formation by the following detergent.
\[\ce{CH3(CH2)10CH2OS\overset{-}{O}3\overset{+}{N}a}\]
Match structures given in Column I with the type of detergents given in Column II.
| Column I | Column II |
| (i) \[\ce{CH3(CH2)16COO(CH2CH2O) nCH2CH2OH}\] | (a) Cationic detergent |
| (ii) \[\ce{C17H35COO- Na+}\] | (b) Anionic detergent |
| (iii) \[\ce{CH3-(CH2)10CH2SO3- Na+}\] | (c) Nonionic detergent |
|
(iv) |
(d) Soap |
Synthetic detergents have advantage over usual soaps as far as cleansing power is concerned. But use of synthetic detergents over a long time creates environmental pollution. How can the pollution caused by synthetic detergents be minimised? Classify the detergents according to their chemical nature.
Explain the following term with suitable example.
Cationic detergents
Explain the following term with suitable examples.
Cationic detergents
Explain the following term with suitable example cationic detergents.
Explain the Following Term with Suitable Examples.
Cationic Detergents
Explain the following term with suitable example.
Cationic detergents
Explain the following term with suitable example:
cationic detergents

