Advertisements
Advertisements
Question
Isostructural species are those which have the same shape and hybridisation. Among the given species identify the isostructural pairs.
Options
\[\ce{[NF3 and BF3]}\]
\[\ce{[BF^{–}4 and NH^{+}4]}\]
\[\ce{[BCl3 and BrCl3]}\]
\[\ce{[NH3 and NO^{-}3]}\]
Advertisements
Solution
\[\ce{[BF^{–}4 and NH^{+}4]}\]
Explanation:
In a molecular structure if a central atom has the same hybridization as the central atom of another molecular species, then the two structures are known as isostructural species.
(i) In \[\ce{NF3}\] the central nitrogen atom is sp3 hybridized and in \[\ce{BF3}\] the central boron atom is sp2. So, these two molecules are not isostructural pairs.
(ii) In \[\ce{BF4^{-}4}\] the central boron atom is sp3 hybridized and in \[\ce{NH^{+}4}\] the central nitrogen atom is also sp3 hybridized. So, these two molecules are identified as isostructural pairs.
(iii) In \[\ce{BCl3}\] the central boron atom is sp2 hybridized and in \[\ce{BrCl3}\] the central bromine atom is sp3d hybridized. So, these two molecules are not isostructural pairs.
(iv) In \[\ce{NH3}\] the central nitrogen atom is sp3 and in \[\ce{NO^{-}3}\] the central nitrogen atom is sp2 hybridized. So, these two molecules are not isostructural pairs.
APPEARS IN
RELATED QUESTIONS
Apart from tetrahedral geometry, another possible geometry for CH4 is square planar with the four H atoms at the corners of the square and the C atom at its centre. Explain why CH4 is not square planar?
Draw a diagram showing the formation of a double bond and a triple bond between carbon atoms in C2H4 and C2H2 molecules.
What is the total number of sigma and pi bonds in the following molecules?
C2H2
What do you understand by bond pairs and lone pairs of electrons? Illustrate by giving one example of each type.
Distinguish between a sigma and a pi bond.
What is the total number of sigma and pi bonds in the following molecules?
C2H4
Which of the following angle corresponds to sp2 hybridisation?
Match the species in Column I with the type of hybrid orbitals in Column II.
| Column I | Column II |
| (i) \[\ce{SF4}\] | (a) sp3d2 |
| (ii) \[\ce{IF5}\] | (b) d2sp3 |
| (iii) \[\ce{NO^{+}2}\], | (c) sp3d |
| (iv) \[\ce{NH^{+}4}\], | (d) sp3 |
| (e) sp |
Match the shape of molecules in Column I with the type of hybridisation in Column II.
| Column I | Column II |
| (i) Tetrahedral | (a) sp2 |
| (ii) Trigonal | (b) sp |
| (iii) Linear | (c) sp3 |
What is the type of hybridisation of carbon atoms marked with star.
\[\begin{array}{cc}
\phantom{.....}\ce{O}\\
\phantom{.....}||\\
\ce{\overset{∗}{C}H2 = CH - \overset{∗}{C} - O - H}
\end{array}\]
What is the type of hybridisation of carbon atoms marked with star.
\[\ce{CH3 - \overset{∗}{C}H2 - OH}\]
What is the type of hybridisation of carbon atoms marked with star.
\[\begin{array}{cc}
\phantom{..........}\ce{O}\\
\phantom{..........}||\\
\ce{CH3 - CH2 - \overset{∗}{C} - H}
\end{array}\]
What is the type of hybridisation of carbon atoms marked with star.
\[\ce{\overset{∗}{C}H3 - CH = CH - CH3}\]
What is the type of hybridisation of carbon atoms marked with star.
\[\ce{CH3 - \overset{∗}{C} ≡ CH}\]
In the given reaction,
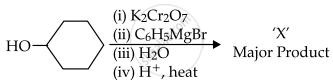
the number of sp2 hybridised carbon (s) in compound 'X' is ______.
The hybridisation of carbanion is:
