Advertisements
Advertisements
Question
In the following complex ion, explain the type of hybridisation, shape and magnetic property:
[Fe(H2O)6]2+
(At. Nos.: Fe = 26)
Long Answer
Advertisements
Solution
The electronic configuration of Fe is [Ar] 4s2 3d6. After the electronic configuration loses two electrons. H2O is a weak field ligand, meaning it does not cause the pairing of electrons in the metal ion. Therefore, in this complex, Fe2+ will have its electrons arranged with unpaired electrons in the d-orbitals.
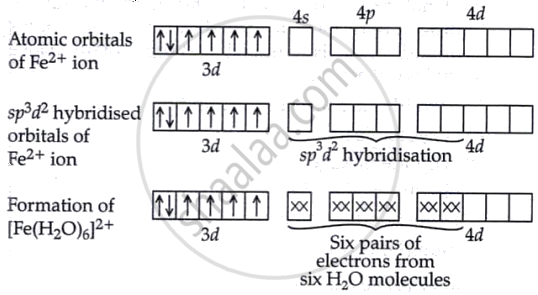
The complex has sp3d2 hybridisation due to the coordination number of 6. This leads to an octahedral shape. Since H2O is a weak field ligand, it does not cause electron pairing, so the Fe2+ complex has unpaired electrons. Therefore, [Fe(H2O)]2+ is paramagnetic.
shaalaa.com
Is there an error in this question or solution?
2024-2025 (March) Delhi Set 3
