Advertisements
Advertisements
Question
In the following complex ion, explain the type of hybridisation, shape and magnetic property:
[NiCl4]2−
(At. Nos.: Ni = 28)
Advertisements
Solution
The electronic configuration for Ni is [Ar] 3d8 4s2. After the loss of two electrons during electronic configuration. Cl is a weak field ligand, indicating that it does not induce electron pairing within the metal ion. Therefore, Ni2+ in this complex will possess its electrons arranged such that unpaired electrons occupy the d-orbitals.
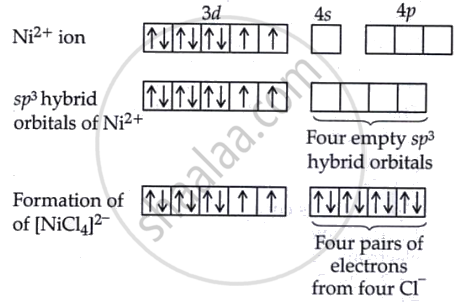
Nickel in Ni2+ undergoes sp3 hybridisation because the coordination number is 4 due to the four Cl ligands. This leads to a tetrahedral molecular geometry. Since Cl is a feeble field ligand, it does not induce electron pairing in the metal ion. Therefore, Ni2+ possesses two unpaired electrons in its 3d electron configuration. As a result, the complex is paramagnetic in nature.
