Advertisements
Advertisements
Question
Identify the order of reaction and write its integrated rate equation, mentioning what each term in the equation represents.
Classify
Equation/Formula
Very Short Answer
Advertisements
Solution
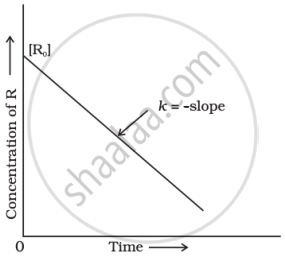
It is a zero-order reaction.
k = `([R] - [R]_0)/t`
Here, k is the rate constant, [R] is the concentration of reactant at time t, and [R0] is the initial concentration of reactant.
shaalaa.com
Is there an error in this question or solution?
