Advertisements
Advertisements
प्रश्न
Identify the order of reaction and write its integrated rate equation, mentioning what each term in the equation represents.
वर्गीकरण करा
समीकरण/सूत्र
अति संक्षिप्त उत्तर
Advertisements
उत्तर
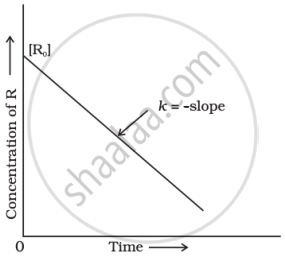
It is a zero-order reaction.
k = `([R] - [R]_0)/t`
Here, k is the rate constant, [R] is the concentration of reactant at time t, and [R0] is the initial concentration of reactant.
shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
