Advertisements
Advertisements
Question
Give two examples of ambidentate ligands.
Advertisements
Solution
- Nitrite (\[\ce{NO^-2}\])
- Thiocyanate (SCN−)
APPEARS IN
RELATED QUESTIONS
Give the oxidation state, d-orbital occupation and coordination number of the central metal ion in the following complex:
K3[Co(C2O4)3]
Give the oxidation state, d-orbital occupation and coordination number of the central metal ion in the following complex:
(NH4)2[CoF4]
Which of the following compounds is used as a semipermeable membrane?
(a) Potassium ferrocyanide
(b) Potassium argentocyanide
(c) Sodium meta aluminate
(d) Copper ferrocyanide
IUPAC name of the following compound is
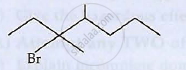
(a) 3 - Bromo- 3, 4- dimethylheptane
(b) 3, 4- dimethyl - 3- bromoheptane
(c) 5- Bromo- 4, 5- dimethylheptane
(d) 4, 5- dimethyl- 5- bromoheptane
Write IUPAC names of the following compounds:

How is Benzonitrile converted to Benzophenone?
Following compounds are given to you :
2-Bromopentane, 2-Bromo-2-methylbutane, 1-Bromopentane
1) Write the compound which is most reactive towards SN2 reaction.
2) Write the compound which is optically active.
3) Write the compound which is most reactive towards β-elimination reaction.
Write the IUPAC name of the following coordination compound:
[PtCl2(NH3)4][PtCl4]
Write structures of compounds A and B of the following reaction :

Ligand (en) is an example of ___________.
In which of the following compounds the oxidation state of the nickel atom is 0?
When 1 mol \[\ce{CrCl3.6H2O}\] is treated with excess of \[\ce{AgNO3}\], 3 mol of \[\ce{AgCl}\] are obtained. The formula of the complex is ______.
A coordination compound \[\ce{CrCl3.4H2O}\] precipitates silver chloride when treated with silver nitrate. The molar conductance of its solution corresponds to a total of two ions. Write structural formula of the compound and name it.
Match the coordination compounds given in Column I with the central metal atoms given in Column II and assign the correct code:
| Column I (Coordination Compound) | Column II (Central metal atom) |
| A. Chlorophyll | 1. rhodium |
| B. Blood pigment | 2. cobalt |
| C. Wilkinson catalyst | 3. calcium |
| D. Vitamin B12 | 4. iron |
| 5. magnesium |
The co-ordinate number and the oxidation state of the element E in the complex [E(en)2(C2O4)]NO2 are respectively?
The most stable ion is ______.
What are Heteroleptic complexes?
Explain the following, giving two examples:
Homoleptic
Give two examples of didentate ligands.
