Advertisements
Advertisements
Question
Give one example of the reaction where order and molecularity are the same.
Mention any two factors that influence the rate of chemical reaction.
If for the reaction A → products, a straight line graph passing through origin is obtained between the rate of reaction against concentration of A, what would be the order of reaction? Why?
Numerical
Advertisements
Solution
- \[\ce{2NO_{2(g)} -> 2NO_{2(g)} + O_{2(g)}}\]
The order of a reaction is 2 and its molecularity is also 2. - Temperature, use of catalyst
- The reaction would be first order.
The differential rate law for the first-order reaction A → products is
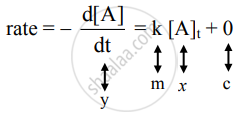
The equation is of the form y = mx + c. A plot of rate versus [A]t is a straight line passing through the origin. The slope of straight line = k.
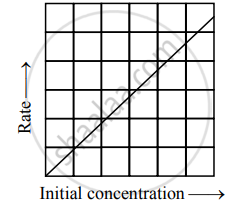
Variation of rate with [A]
shaalaa.com
Is there an error in this question or solution?
