Advertisements
Advertisements
Question
Give a chemical test to distinguish between the following pair of compounds:
Hydrogen chloride gas and hydrogen sulphide gas
Advertisements
Solution
When exposed to hydrogen sulphide gas, lead acetate paper that is moist turns black; when exposed to hydrogen chloride gas, it remains colourless.
RELATED QUESTIONS
Explain why anhydrous HCl is a poor conductor while aqueous HCl is an excellent conductor.
Write an equation for the reaction of hydrochloric acid on copper oxide.
Name the following:
A chemical in which gold can be dissolved.
How will you distinguish between the two black samples, CuO and MnO2 with a chemical test?
Identify the anion present of the following compound:
When barium chloride solution is added to salt solution E a white precipitate insoluble in dilute hydrochloric acid is obtained
Write the balanced equation for the reaction of dilute hydrochloric acid with the following:
Iron (II) sulphide
A solution of hydrogen chloride in water is prepared. The following substances are added to separate portions of the solution :
| S.NO. | Substances added | Gas evolved | Odour |
| 1. | Calcium carbonate | ______ | ______ |
| 2. | Magnesium ribbon | ______ | ______ |
| 3. | Manganese (IV) oxide with heating | ______ | ______ |
| 4. | Sodium sulphide | ______ | ______ |
Complete the table by writing the gas evolved in each case and its odour.
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D, and E.
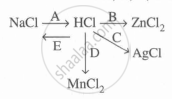
State which of the two - a solution of HCl in water or in toluene is an electrolyte. Explain.
The ratio between the volumes occupied by 4.4 grams of carbon dioxide and 2 grams of hydrogen gas is ______.
