Advertisements
Advertisements
Question
Give a balanced equation for –
A displacement reaction involving a metal above hydrogen in the activity series with copper [II] sulphate solution
Advertisements
Solution
Displacement reaction
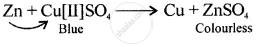
[more reactive displaces less reactive Cu]
APPEARS IN
RELATED QUESTIONS
In the refining of silver, the recovery of silver from silver nitrate solution involved displacement by copper metal. Write down the reaction involved.
What type of chemical reaction is represented by the following equation?
\[\ce{A + BC -> AC + B}\]
Which of the following is a combination and which is a displacement reaction?
- \[\ce{Cl2 + 2KI -> 2KCl + I2}\]
- \[\ce{2K + Cl2 -> 2KCl}\]
What happens when a piece of iron metal is placed in copper sulphate solution? Name the type of reaction involved.
What happens when a zinc strip is dipped into a copper sulphate solution?
Name the type of reaction involved.
A strip of metal X is dipped in a blue coloured salt solution YSO4. After some time, a layer of metal Y from the salt solution is formed on the surface of metal strip X. Metal X is used in galvanisation whereas metal Y is used in making electric wires. Metal X and metal Y together form an alloy Z.
- What could metal X be?
- What could metal Y be?
- Name the metal salt YSO4.
- What type of chemical reaction takes place when metal X reacts with salt solution YSO4? Write the equation of the chemical reaction involved.
- Name the alloy Z.
When a strip of red-brown metal X is placed in a colourless salt solution YNO3 then metal Y is set free and a blue coloured salt solution X(NO3)2 is formed. The liberated metal Y forms a shining white deposit on the strip of metal Х.
- What do you think metal X is?
- Name the salt YNO3.
- What could be metal Y?
- Name the salt X(NO3)2.
- What type of reaction takes place between metal X and salt solution YNO3?
Fill in the blank.
The chemical change involving iron and hydrochloric acid illustrates a ...............reaction.
Complete the following chemical reaction:
\[\ce{CuSO4 (aq) + Pb (s) -> \underline{}\underline{}\underline{}\underline{}\underline{}\underline{} + \underline{}\underline{}\underline{}\underline{}\underline{}\underline{}}\]
Name the chemical reaction type.
State the change in colour observed in following case mentioning the reason:
A piece of zinc is dipped in ferrous sulphate solution.
