Advertisements
Advertisements
प्रश्न
Give a balanced equation for –
A displacement reaction involving a metal above hydrogen in the activity series with copper [II] sulphate solution
Advertisements
उत्तर
Displacement reaction
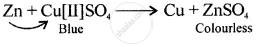
[more reactive displaces less reactive Cu]
APPEARS IN
संबंधित प्रश्न
CuSO4(aq) + Zn(s) → ZnSO4(aq) + Cu(s) is an example of a decomposition reaction.
Write the balanced chemical equation for the following and identify the type of reaction.
\[\ce{Magnesium(s) + Hydrochloric acid(aq) -> Magnesium chloride(aq) + Hydrogen(g)}\]
Why does the colour of copper sulphate solution change when an iron nail is kept immersed in it?
What is meant by a displacement reaction? Explain with the help of one example.
What happens when a piece of iron metal is placed in copper sulphate solution? Name the type of reaction involved.
Explain the following type of chemical reaction, giving two examples for it:
Displacement reaction
What is observed after about 1 hour of adding the strips of copper and aluminium separately to the ferrous sulphate solution filled in two beakers? Name the reaction if any change in colour is noticed. Also, write a chemical equation for the reaction.
- Complete the given chemical reaction:
\[\ce{CuSO4_{(aq)} + Fe_{(s)} ->}\] _______ + _______ - Name the type of the reaction.
Match the following
Identify the types of reactions.
| REACTION | TYPE |
| NH4OH(aq) + CH3COOH(aq) → CH3COONH4(aq) + H2O(l) | Single Displacement |
| Zn(s) + CuSO4(aq) → ZnSO4(aq) + Cu(s) | Combustion |
| \[\ce{ZnCO3_{(s)} + ->[Heat]ZnO_{(s)} + CO2_{(g)}}\] | Neutralization |
| C2H4(g) + 4O2(g) → 2CO2(g) + 2H2 O(g) + Heat | Thermal decomposition |
State the change in colour observed in following case mentioning the reason:
A piece of zinc is dipped in ferrous sulphate solution.
