Advertisements
Advertisements
Question
For the decomposition of azomethane
\[\ce{CH3N2CH3_{(g)} -> CH3CH3_{(g)} + N2_{(g)}}\] is a first order reaction, the variation in partial pressure with time at 600 K is given by
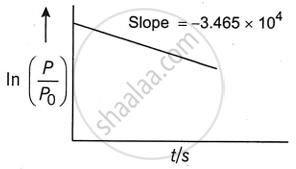
The half-life of the reaction is ______ × 10−5 s (Nearest integer).
Fill in the Blanks
Advertisements
Solution
The half-life of the reaction is 2 × 10−5 s.
Explanation:
For a first-order reaction, the rate law is given by:
`ln(P/P^circ) = -kt`
So the slope of the straight line = −k.
Thus,
k = 3.465 × 104 s−1 ...[Given]
The half-life t1/2 for a first-order reaction is given by:
`t_(1//2) = (ln 2)/k`
= `0.693/(3.465 xx 10^4)`
= `0.693/34650`
= 2 × 10−5 s
∴ The half-life of the reaction is 2 × 10−5 s.
shaalaa.com
Is there an error in this question or solution?
