Advertisements
Advertisements
Question
For each of the salt: A, B, C and D, suggest a suitable method of its preparation.
C is a soluble salt of copper.
Advertisements
Solution
Copper sulphate [CuSO4.5H2O]. By the action of acid on metal oxide.
RELATED QUESTIONS
Write a word equation and then a balanced equation for the reaction taking place when:
Dilute hydrochloric acid reacts with magnesium ribbon.
Complete and balance the following chemical equations:
CuO (s) + HCI (aq) →
Give chemical equation of the reaction involved.
Write a balanced chemical equation of the reaction which takes place.
What would be the colour of litmus in a solution of sodium carbonate?
Write the chemical formula of washing soda. How can it be obtained from baking soda? List two industries in which washing soda is used for other purposes than washing clothes.
Egg shell is made up of ____________.
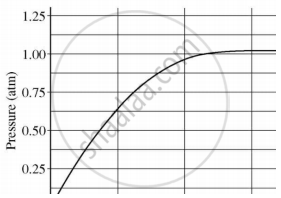
A student added 10 g of calcium carbonate in a rigid container, secured it tightly and started to heat it. After some time, an increase in pressure was observed, the pressure reading was then noted at intervals of 5 mins and plotted against time, in a graph as shown below. During which time interval did maximum decomposition take place?
Solutions of acids conduct ______.
Give a balanced equation for the reaction:
Silver nitrate solution and sodium chloride solution.
