Advertisements
Advertisements
Question
Fill in the blank
Chalk powder dissolved in water is an example of a Chalk powder dissolved in water is an example of a _________.
Advertisements
Solution
Chalk powder dissolved in water is an example of a Chalk powder dissolved in water is an example of a suspension.
APPEARS IN
RELATED QUESTIONS
Classify the following substances into compounds and mixtures:
Fill in the blan
An ________ is a homogeneous mixture of two or more metals.
Name the substance you will add to speed up sedimentation.
Give one example for the following types of mixtures:
liquid-gas homogenous mixture
Name: a Non-metal Which is a Good Conductor of Electricity.
Give two examples of the following.
Metals which are soft
State the number of atoms of each element, present in \[\ce{HNO3}\].
Also name the compound.
Give one use of the following substance:
Coal
Answer the following question:
What do you understand by the statement: ‘metals are ductile and malleable’?
Indicate whether the following statement is true or false:
All elements are artificially prepared
Tick (√) the correct alternative from the choice given for the following statement
The hardest naturally occurring substance
The important physical properties of substances are colour, odour, nature, density & solubility in water.
Name - a poisonous gas almost as heavy as air.
Match the statements in List I – 1 to 10 correct answers in List II-A to J.
| List I | LIst II | ||
| 1 | Fluorine and chlorine | A | Nitrogen |
| 2 | A diatomic molecule | B | Boron |
| 3 | A molecule containing the elements hydrogen and oxygen. | C | Reactants |
| 4 | A metalloid - showing properties of both metals and non-metals. | D | Chemical Formula |
| 5 | The term used for the substances which take part in the chemical reaction | E | Halogens |
| 6 | The term represents a substance by means of symbols. | F | Water |
| 7 | The term used for the substances formed as a result of a chemical reaction | G | Nitric Acid |
| 8 | A compound containing one atom of zinc and two atoms of chlorine. | H | Products |
| 9 | The chemical name for nitrogen monoxide. | I | Phosphorus |
| 10 | A polyatomic molecule. | J | Zinc Chloride |
Who are my companions?
| Group 'A' | Group 'B' |
| 1. Stainless steel | (a) Non-metal |
| 2. Silver | (b) compound |
| 3. Bhajani mixture for milling | (c) Mixture |
| 4. Salt | (d) Element |
| 5. Coal | (e) Alloy |
| 6. Hydrogen | (f) Metal |
Write the names of elements from the following symbol:
Cd
Write an answer to the following question in your own words.
Which elements (metals and non-metals), compounds and mixtures do we use in our day-to-day life?
Select the correct answer to the statement given below:
The chemical name for dinitrogen oxide [N20].
Define the terms elements, compounds & mixtures with a view to show their basic difference.
The first letter of an element is always written in ______ letter.
Higher Order Thinking Skills.
In the diagram given below, the circle, square and triangle represent the atoms of different elements.
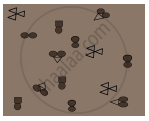
Identify all combinations that represent
- molecule of a compound
- molecule of an element consisting of two atoms
- molecule of an element consisting of three atoms
