Advertisements
Advertisements
Question
Explain why high pressure is required in the manufacture of sulphur trioxide by the contact process. State the law or principle used.
Explain
Law
Advertisements
Solution 1
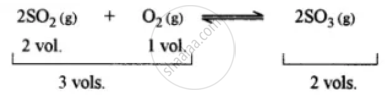
This is because the forward reaction is accompanied by a decrease in volume. The principle used is, i.e., Chatelier’s principle. It states that when a system in equilibrium is subjected to stress (i.e., change of concentration, temperature, pressure, etc.), the equilibrium tends to shift in a direction so as to undergo the effect of applied stress.
shaalaa.com
Solution 2
- High pressure is required because the reaction to form sulphur trioxide from sulphur dioxide and oxygen involves a decrease in the number of gas moles (from 3 to 2).
- Increasing pressure shifts the equilibrium towards the product side, increasing SO3 yield.
- The principle used is Le-Chatelier’s Principle, which states that a system shifts equilibrium to counteract a change (here, pressure increase favours fewer moles).
- In practice, about 2 atmospheres of pressure is maintained to favour SO3 formation.
shaalaa.com
Is there an error in this question or solution?
