Advertisements
Advertisements
Question
Explain the structure of a typical antibody molecule.
Advertisements
Solution
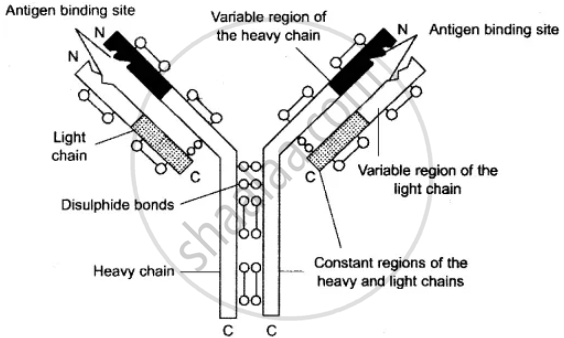
Structure of an antibody is a glycoprotein, called immunoglobulin, that has a specific amino acid sequence by which it can interact with specific antigens. Antibodies form 20% of the plasma proteins. Each antibody combines at least 2 light (L) and 2 heavy (H) polypeptide chains. The heavy chains have a larger number of amino acids, while the lighter chain has a smaller number of them. Usually, the polypeptides form a Y-shaped configuration. The stem of Y is exclusively formed by heavy chains. In the arms of Y, both light and heavy chains occur parallel to each other except for antigen-binding sites. Attachments and bendings occur by means of disulphide bonds (S-S). In certain immunoglobulins, the number of chain pairs can be JO. An antibody has a variable portion in the arms, known as the V-region or antigen-binding fragment (Fab), which allows it to bind to specific antigens. The remainder of the antibody is called the constant portion, which is also known as the crystalline fragment, Fc, and it remains the same across different antibodies.
Most of the antibodies function as monomers. A few, such as IgA and IgM, can occur both as monomers and polymers.
