Advertisements
Advertisements
Question
Explain the meaning of heat and work with suitable examples.
Advertisements
Solution
Meaning of heat: When an object at a higher temperature is placed in contact with another object at a lower temperature, there will be a spontaneous flow of energy from the object at a higher temperature to the one at a lower temperature. This energy is called heat. This process of energy transfer from higher temperature objects to lower temperature objects is called heating. Due to the flow of heat sometimes the temperature of the body will increase or sometimes it may not increase.
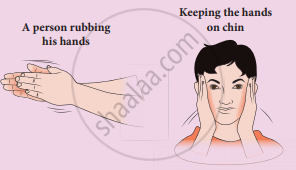
Meaning of work: When you rub your hands against each other the temperature of the hand's increases. You have done some work on your hands by rubbing. The temperature of the hands increases due to this work. Now if you place your hands on the cheek, the temperature of the cheek increases. This is because the hands are at a higher temperature than the cheek. In the above example, the temperature of the hands is increased due to work, and the temperature of the cheek is increased due to heat transfer from the hands to the chin. It is shown in the Figure. By doing work on the system, the temperature in the system will increase and sometimes may not. Like heat, work is also not a quantity and through the work-energy is transferred to the system. So we cannot use the word ‘the object contains more work’ or ‘less work’.
Either the system can transfer energy to the surrounding by doing work on surrounding or the surrounding may transfer energy to the system by doing work on the system. For the transfer of energy from one body to another body through the process of work, they need not be at different temperatures.
APPEARS IN
RELATED QUESTIONS
The kinetic energy per molecule of a gs at temperature T is ________.
(a) `(3/2)RT`
(b) `(3/2)K_BT`
(c) `(2/3) RT`
(d) `(3/2)("RT"/M)`
The density of water at 4°C is supposed to be 1000 kg m–3. Is it same at sea level and at high altitude?
In a calorimeter, the heat given by the hot object is assumed to be equal to the heat taken by the cold object. Does it mean that heat of the two objects taken together remains constant?
Should a thermometer bulb have large heat capacity or small heat capacity?
Temperature and Heat are ______
One day in 1922, the air temperature was measured at 59°C in the shade in Libya ______.
Analogy
Evaporation:: 100°C: Freezing:: ______.
An earthen pitcher loses 1 gm of water per minute due to evaporation. If the water equivalent of the pitcher is 0.5 kg and the pitcher contains 9.5 kg of water, calculate the time required for the water in a pitcher to cool to 28°C from the original temperature of 30°C. Neglect radiation effects. The latent heat of vaporization in this range of temperature is 580 Cal/gm and the specific heat of water is 1 Cal/gm°C.
The temperature of a liquid drops from 365 K to 361 K in 2 min. Find the time during which the temperature of the liquid drops from 344 K to 342 K.
(Take, room temperature = 293 K)
Which formula gives the average kinetic energy per molecule of an ideal gas?
