Advertisements
Advertisements
Question
Explain the rule with example according to which electrons are filled in various energy levels
Advertisements
Solution
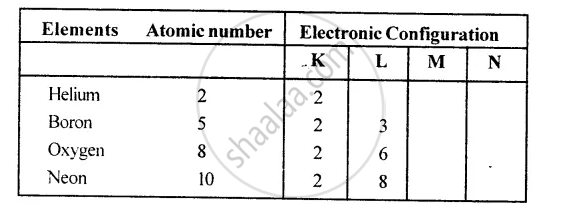
The maximum number of electrons that can be present in any shell or orbit of an atom is given by the formula 2n2, where n is the serial number of the shell.
Therefore:
K shell, n = 1, no. of electrons = 2 x 12 = 2
L shell, n = 2, no. of electrons = 2 x 22 = 8
M shell, n = 3, no. of electrons = 2 x 32 = 18
N shell, n = 4, no. of electrons = 2 x 42= 32
Electrons are not accommodated in a given shell, unless the inner shells are filled.
That is, the shells are filled in a stepwise manner
APPEARS IN
RELATED QUESTIONS
Give the following a suitable word/phrase
The sub-atomic particle with negative charge and negligible mass.
Give the following a suitable word/phrase.
The sum of the number of protons and neutrons of an atom.
Multiple Choice Questions
The outermost shell of an atom is known as
What are the two main parts of which an atom is made of?
ANSWER IN DETAIL
What is the maximum number of electrons that L shell can accommodate ? Give reasons for your answer.
Name the sub-atomic particle whose charge is 0.
Name the three fundamental particles of an atom.
Explain how the modern atomic theory contradicted Dalton’s atomic theory.
Draw a neat labelled diagram representing an atom. Name the three sub-atomic particles in the atom and represent them symbolically showing the mass and charge of each. State where the sub-atomic particles are present in the atom.
If an atom ‘A’ has an atomic number of eleven, state the number of protons and electrons it contains.
