Advertisements
Advertisements
प्रश्न
Explain the rule with example according to which electrons are filled in various energy levels
Advertisements
उत्तर
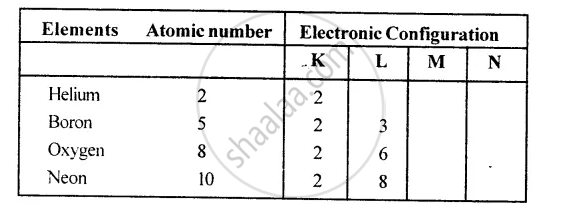
The maximum number of electrons that can be present in any shell or orbit of an atom is given by the formula 2n2, where n is the serial number of the shell.
Therefore:
K shell, n = 1, no. of electrons = 2 x 12 = 2
L shell, n = 2, no. of electrons = 2 x 22 = 8
M shell, n = 3, no. of electrons = 2 x 32 = 18
N shell, n = 4, no. of electrons = 2 x 42= 32
Electrons are not accommodated in a given shell, unless the inner shells are filled.
That is, the shells are filled in a stepwise manner
APPEARS IN
संबंधित प्रश्न
Give the following a suitable word/phrase.
The electrons present in the outermost shell.
Multiple Choice Questions
The outermost shell of an atom is known as
Tick the most appropriate answer.
Which of the following scientists observed that cathode rays consists of negatively charged particles?
- John Dalton
- J J Thomson
- James Chadwick
- Democritus
WRITE SHORT ANSWER
Name the isotopes of hydrogen.
State whether the following statement is true or false :
The anode rays obtained from all the gases consist of positively charged particles called protons.
Define the term Mass number.
Define the term Nucleons.
Explain how the modern atomic theory contradicted Dalton’s atomic theory.
Draw a neat labelled diagram representing an atom. Name the three sub-atomic particles in the atom and represent them symbolically showing the mass and charge of each. State where the sub-atomic particles are present in the atom.
Write down the names of the particles represented by the following symbols and explain the meaning of superscript and subscript numbers attached.
`""_1"H"^1`, `""_0"n"^1`,`""_(-1)"e"^0`
