Advertisements
Advertisements
Question
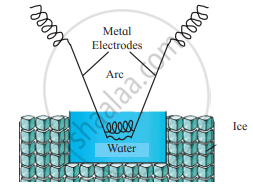
Draw labelled diagram of Bredig’s arc method.
Advertisements
Solution
APPEARS IN
RELATED QUESTIONS
Define the following with a suitable example, of each:
Multimolecular colloid
Identify the dispersed phase and dispersion medium in the following colloidal dispersions.
- milk
- blood
- printing ink
- fog
Write a note on the Tyndall effect.
Draw labelled diagram of Soap micelle.
Which one of the following methods is used to make platinum sol?
Which of the following is multimolecular colloid?
In coagulation of positively charged ferric hydroxide sol, the most and least effective ions are ____________ and ____________ respectively.
Which of the following is NOT true for lyophilic colloids?
Smoke is an example of ____________.
Froth is a colloidal solution of ____________.
Which of the following is multimqlecular colloid?
Tyndall effect is useful ______.
Some colloids are stable by their nature, i.e., gels, alloys, and solid foams. Gelatin and jellies are two common examples of a gel. The solid and liquid phases in a gel are interdispersed with both phases being continuous. In most systems, the major factor influencing the stability is the charge on the colloidal particles. If a particular ion is preferentially adsorbed on the surface of the particles, the particles in suspension will repel each other, thereby preventing the formation of aggregates that are larger than colloidal dimensions. The ion can be either positive or negative depending on the particular colloidal system, i.e., air bubbles accumulate negative ions, sulphur particles have a net negative charge in a sulphur sol, and the particles in a metal hydroxide sol are positively charged. Accumulation of charge on a surface is not an unusual phenomenon-dust is attracted to furniture surfaces by electrostatic forces. When salts are added to lyophobic colloidal systems the colloidal particles begin to form larger aggregates and a sediment forms as they settle. This phenomenon is called flocculation, and the suspension can be referred to as flocculated, or colloidally unstable. If the salt is removed, the suspension can usually be restored to its original state; this process is called deflocculation or peptization. The original and restored colloidal systems are called deflocculated, peptized, or stable sols.
Why does a small amount of salt have such a dramatic effect on the stability of a lyophobic colloidal system? The answer lies in an understanding of the attractive and repulsive forces that exist between colloidal particles. Van der Waals forces are responsible for the attractions, while the repulsive forces are due to the surface charge on the particles. In a stable colloid, the repulsive forces are of greater magnitude than the attractive forces. The magnitude of the electrical repulsion is diminished by addition of ionized salt, which allows the dispersed particles to aggregate and flocculate. River deltas provide an example of this behaviour. A delta is formed at the mouth of a river because the colloidal clay particles are flocculated when the freshwater mixes with the salt water of the ocean.
Gelatin is a _________ colloidal system.
Which of the following electrolytes will have maximum coagulating value for AgI/Ag+ solution?
A colloidal system having a solid substance as a dispersed phase and a liquid as a dispersion medium is classified as ______.
Which of the following substances will precipitate the negatively charged emulsions?
(i) \[\ce{KCl}\]
(ii) glucose
(iii) urea
(iv) \[\ce{NaCl}\]
On the basis of Hardy-Schulze rule explain why the coagulating power of phosphate is higher than chloride.
Match the items of Column I and Column II.
| Column I | Column II |
| (i) Butter | (a) dispersion of liquid in liquid |
| (ii) Pumice stone | (b) dispersion of solid in liquid |
| (iii) Milk | (c) dispersion of gas in solid |
| (iv) Paints | (d) dispersion of liquid in solid |
Cloud is an example of
The coagulation of 200 ML of position sol took place when 0.73 HCL was added to its without changing the volume much. The flocculation value of HCL for the colloid is.
Which of the following substances will precipitate the negatively charge emulsion
Which of the following electrolytes will be most effective in coagulating negative sol?
Blood may be purified by ______.
Identify the colloidal molecule containing hydrophilic head and hydrophobic tail within it.
Identify positively charged sol from following.
Identify the correct decreasing order of precipitation power of flocculating ion added, from following.
