Advertisements
Advertisements
Question
Differentiate between the following pair based on the criteria given in the bracket:
Sulphuric acid and hydrochloric acid [type of salts formed]
Advertisements
Solution
| Sulphuric Acid | Hydrochloric Acid |
| Reacts with bases to form sulphate salts on neutralization. | Reacts with bases to form chloride salts on neutralization. |
APPEARS IN
RELATED QUESTIONS
State which of the two a solution of HCl in water or in toluene is an electrolyte. Explain.
Write an equation for the reactions of hydrochloric acid on manganese (IV) oxide.
State which of the two - a solution of HCl in water or in toluene is an electrolyte. Explain.
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.
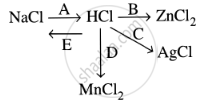
Write an equation for the reaction of hydrochloric acid on manganese (IV) oxide.
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.
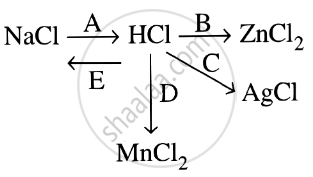
State which of the two — a solution of HCl in water or in toluene is an electrolyte. Explain.
Convert Hydrochloric acid to nascent chlorine.
Write an equation for the reaction of hydrochloric acid on manganese (IV) oxide.
State which of the two - a solution of HCl in water or in toluene is an electrolyte. Explain.
