Advertisements
Advertisements
Question
Describe the reaction that show
Concentrated sulphuric acid is a non-volatile acid.
Advertisements
Solution
Concentrated sulphuric acid is non-volatile hence it displaces more volatile acids.
\[\ce{NaCl + H2SO4 ->[200°C] NaHSO4 + HCl}\]
APPEARS IN
RELATED QUESTIONS
State one relevant observation for given reactions:
Addition of ethyl alcohol to acetic acid in the presence of concentrated Sulphuric acid
Write balanced chemical equations for the action of dilute sulphuric acid on sodium sulphite.
Give balanced chemical equations for the action of sulphuric acid on the following:
Potassium hydrogen carbonate.
Distinguish between the given pair of compounds using the test given within brackets:
Sodium nitrate and sodium sulphite (using dilute sulphuric acid).
Give a chemical test to distinguish between dilute sulphuric acid and conc. sulphuric acid.
Write balanced equation for the reaction of dilute sulphuric acid with the following:
Lead nitrate solution
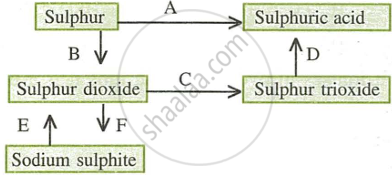
- Name the catalyst which helps in the conversion of sulphur dioxide to sulphur trioxide in step C.
- In the contact process for the manufacture of sulphuric acid, sulphur trioxide is not converted to sulphuric acid by reacting it with water. Instead a two-step procedure is used. Write the equations for the two steps involved in D.
- What type of substance will liberate sulphur dioxide from sodium sulphite in step E?
- Write the equation for the reaction by which sulphur dioxide is converted to sodium sulphite in step F.
Name the gas evolved in following case:
The gas produced by the action of concentrated sulphuric acid on sodium chloride.
Write the observation for the following:
Decomposition of bicarbonates by dil. H2SO4.
\[\ce{2NaHCO3 + H2SO4 -> Na2SO4 + 2H2O + 2CO2}\]
Hydrogen chloride gas is prepared in the laboratory by the action of concentrated sulphuric acid on sodium chloride.
Give a balanced chemical equation for the above reaction.
