Advertisements
Advertisements
Question
Define one calorie.
Advertisements
Solution
One calorie is the amount of heat energy required to raise the temperature of 1 gram of water through 1°C.
RELATED QUESTIONS
Match the following
| Column A | Column B |
| 1. mercury | 1. insulator |
| 2. wood | 2. water from 0°C to 4°C |
| 3. Aluminum | 3.absorbs |
| 4. contracts | 4.conductor |
| 5.black surface | 5. Thermometer |
Select the correct alternative
A thermometer uses
While deciding the unit for heat, which temperature interval is chosen? Why?
What will absorb heat?
Iron griddle (tava)
What will absorb heat?
Plastic plate
Give scientific reason.
Dew drops form on the grass in winter.
Name the SI unit of heat.
Which temperature segment is chosen when determining the unit of heat? Why?
The unit of heat and temperature are the same.
The process of converting a liquid into a solid is called ________.
Why does the bottom of a lake not freeze in severe winter though the surface is all frozen?
How can heat energy be generated?
A circular metal loop is heated at point O as shown in the following figure.
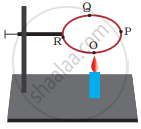
- In which direction would heat flow in the loop?
- In which order are the pins at points P, Q and R fixed with the help of wax fall if points O, P, Q and R are equidistant from each other?
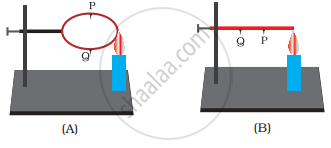
In the arrangements A and B shown in the following figure, pins P and Q are fixed to a metal loop and an iron rod with the help of wax. In which case are both the pins likely to fall at different times? Explain.
The SI unit of heat energy is ______.
The water in the black can becomes ______ than that in a white can after exposure to the sun.
In summer, we prefer light – colored clothes and in winter we usually wear dark – colored clothes.
Match the following
| 1. | Heat | a. | Good absorber |
| 2. | Temperature | b. | Form of energy |
| 3. | Black surface | c. | Insulators |
| 4. | Rubber, cork | d. | The measure of hotness or coldness |
What is heat energy?
Heat gained by a body depends upon which factors?
