Advertisements
Advertisements
प्रश्न
Define one calorie.
Advertisements
उत्तर
One calorie is the amount of heat energy required to raise the temperature of 1 gram of water through 1°C.
संबंधित प्रश्न
Name the mode of transfer of heat in the following :
(a) solid,
(b) liquid,
(c) gas
(d) vacuum
Define the term calorie. How is it related to joule (the S.I. unit of heat)?
Cooking utensils are made from metals due to their property of ______.
What will absorb heat?
Wooden board
What will absorb heat?
Glass
1 calorie is 4.81 joules.
When an object is heated, the molecules that make up the object
A device in which the loss of heat due to conduction, convection and radiation is minimized is ______.
The sun gives us light and heat.
We can absorb heat by rubbing two surfaces of some substances.
In the past, people used to rub two wooden pieces together to light fire.
When an electric current is passing through the conductor ______ energy is produced.
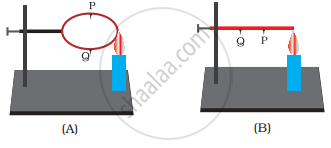
In the arrangements A and B shown in the following figure, pins P and Q are fixed to a metal loop and an iron rod with the help of wax. In which case are both the pins likely to fall at different times? Explain.
Observe the picture given in figure. Water is being boiled in a pan of wide base.

- Which position P or T will feel warmer?
- Fill up the boxes P and T to indicate the mode of flow of heat to the hand.
In summer, we prefer light – colored clothes and in winter we usually wear dark – colored clothes.
Assertion: Small gaps left between railway lines.
Reason: It allows for the contraction of rails during summer.
In which of the following, chemical energy is converted into heat energy?
Match the following:
| Column A | Column B | ||
| 1. | Heat | a. | Heat gained or lost in the change of state without any change in temperature |
| 2. | m × L. | b. | Heat gained or lost when there is no change of state |
| 3. | Temperature | c. | Form of energy |
| 4. | m × s × t | d. | SI unit of specific latent heat |
| 5. | J/Kg | e. | degree of hotness or coldness |
