Advertisements
Advertisements
Question
Copy and complete the following table.
Column 3 has the names of gases to be prepared using the substance you enter in column 1, along with dilute or concentrated sulphuric acid, as indicated in column 2.
| Column 1 | Column 2 | Column 3 |
| Substance reacted with acid | Dilute or concentrated sulphuric acid | Gas |
| Hydrogen | ||
| Carbon dioxide | ||
| Only chlorine |
Write the equation for the laboratory preparation of :
(i) Sodium sulphate using dilute sulphuric acid.
(ii) Lead sulphate using dilute sulphuric acid.
Advertisements
Solution
| Column 1 | Column 2 | Column 3 |
| Substance reacted with acid | Dilute or concentrated sulphuric acid | Gas |
| Metal | Dilute sulphuric acid | Hydrogen |
| Carbonate | Dilute sulphuric acid | Carbon dioxide |
| Metal chlorides | Conc. sulphuric acid | Only chlorine |
Equation are :
(i) \[\ce{2NaOH + H2SO4 -> Na2SO4 + 2H2O}\]
(ii) \[\ce{Pb(NO3)2 + H2SO4 -> PbSO4 v + 2HNO3}\]
APPEARS IN
RELATED QUESTIONS
The acid which is used in the preparation of a volatile acid
Identify the acid in the following case:
The acid which produces sugar charcoal from sugar.
Give a chemical test to distinguish between dilute sulphuric acid and dilute hydrochloric acid.
Name the following:
Products obtained by treating ferrous sulphide with dilute sulphuric acid.
Name the anion [negative ion] present in the following compound:
Compound D when warmed with dilute H2SO4 gives a gas which turns acidified potassium dichromate solution green.
Write the equation for the laboratory preparation of the following salts, using sulphuric acid.
Lead sulphate from lead nitrate.
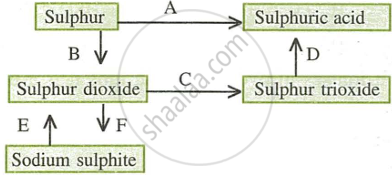
- Name the catalyst which helps in the conversion of sulphur dioxide to sulphur trioxide in step C.
- In the contact process for the manufacture of sulphuric acid, sulphur trioxide is not converted to sulphuric acid by reacting it with water. Instead a two-step procedure is used. Write the equations for the two steps involved in D.
- What type of substance will liberate sulphur dioxide from sodium sulphite in step E?
- Write the equation for the reaction by which sulphur dioxide is converted to sodium sulphite in step F.
In the given equation identify the role played by concentrated sulphuric acid S + 2H2SO4→ 3SO2 + 2H2O
Give two balanced reactions of the type to show the following property of sulphuric acid:
Dehydrating nature
Convert the following reaction into a balanced chemical equation:
Sodium hydroxide to sodium sulphate using sulphuric acid.
