Advertisements
Advertisements
Question
Complete the following flow chart and answer the questions below:

i) In which method pine oil is used?
ii) Explain what method of concentration in brief.
Advertisements
Solution

i. In froth floatation method.
Industrially, pine oil is used as a frother in mineral extraction from ores. For example, in copper extraction, pine oil is used to condition copper sulfide ores for froth flotation. Therefore, it is important in the industry for the froth flotation process
ii. the finely ground ore is put into a big tank containing an ample amount of water. Certain oils such as pine oil, eucalyptus oil, is added in the water for the formation of froth. Due to agitation a foam is formed from oil, water, and air bubbles together and floats on the surface of the water.
1) The Froth Floatation method is a method used for the concentration of metal ores.
2) It is used for the concentration of sulfide ores.
3) In this method, water is added to powdered ore.
4) To this, substances called collectors (such as pine oil ) are added so that the ore particles can be separated from the gangue particles.
5)The ore particles become wet by oil while the gangue particles become wet by water.
6) Air is blown in through a pipe and a rotating paddle agitates this mixture.
7) This forms froth.
8) The froth, being lighter, carries with it the ore particles and is separated from gangue.
9) So, the ore particles float above water. The froth is then skimmed off, leaving behind the gangue.
10) It is dried and metal is thus obtained.
APPEARS IN
RELATED QUESTIONS
Why are alkali metals kept in kerosene oil?
Metal A has an electronic configuration of 2, 8, 1 and metal B has 2, 8, 8, 2 which is more reactive metal.
Give the effect of heat on their: hydroxide
`Ag2O` ................................
................................
`2NaNO_3` ......................
......................
`2Pb(NO3)_2`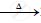 .........................
.........................
`2Cu_2O +Cu_2S` ......................
......................
`HgS + O_2`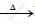 ..................
..................
State two properties of brass that render it more useful for some purpose than its components.
Aluminium is extracted from its chief ore, bauxite. The ore is first purified and then the metal is extracted from it by electrolytic reduction.
Write three balanced equations for the purification of bauxite.
Why does iron or zinc not occur free in nature?
What do you observe when hydrogen is passed over heated copper oxide?
Identify the following method of concentration of ores and explain briefly.
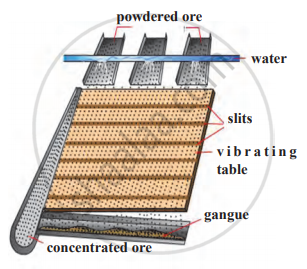
Explain the froth floatation method with a neat labelled diagram.
Complete the following flowchart.

Observe the figure and name and explain in brief the following method:

Calculate the difference between heat of combustion of carbon monoxide gas at constant pressure and at constant volume at 27°C? (R = 2 cal K-1 mol-1).
Limestone is used as a flux in the extraction of ______.
Explain the following term:
Metallurgy
Calcination is used in metallurgy for the removal of?
