Advertisements
Advertisements
Question
Boric acid is polymeric due to ______.
Options
its acidic nature
the presence of hydrogen bonds
its monobasic nature
its geometry
Advertisements
Solution
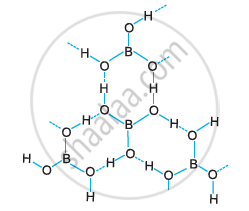
Boric acid is polymeric due to the presence of hydrogen bonds.
Explanation:
Boric acid is polymeric because of the presence of hydrogen bonds. In the given figure, the dotted lines represent hydrogen bonds.
RELATED QUESTIONS
Explain what happens when boric acid is heated.
Explain the Structure of Boric Acid.
Explain the following reaction.
Hydrated alumina is treated with aqueous NaOH solution.
A certain salt X, gives the following results.
- Its aqueous solution is alkaline to litmus.
- It swells up to a glassy material Y on strong heating.
- When conc. H2SO4 is added to a hot solution of X, white crystal of an acid Z separates out.
Write equations for all the above reactions and identify X, Y and Z.
Write a balanced equation for NaH + B2H6 → ?
Boric acid is an acid because its molecule ______.
Draw the structures of BCl3.NH3 and AlCl3 (dimer).
Explain the nature of boric acid as a Lewis acid in water.
Draw the structure of boric acid showing hydrogen bonding. Which species is present in water? What is the hybridisation of boron in this species?
A compound (A) of boron reacts with NMe3 to give an adduct (B) which on hydrolysis gives a compound (C) and hydrogen gas. Compound (C) is an acid. Identify the compounds A, B and C. Give the reactions involved.
Which is correct statement about diborane structure?
Aqueous solution of which of the following boron compounds will be strongly basic in nature?
Diborane (B2H6) reacts independently with O2 and H2O to produce, respectively:
Boric acid heated to red hot gives ______.
On the addition of mineral acid to an aqueous solution of borax, the compound formed is ______.
Which one of the following methods is used to prepare borax crystals?
