Advertisements
Advertisements
Question
At 90°C pure water has [H3O+] = 10−6. What is the value of Kω at this temperature?
Options
10−6
10−12
10−13
10−14
MCQ
Advertisements
Solution
10−12
Explanation:
\[\ce{H2O <=> H^+ + OH^-}\]
Kh = `(["H"^+].["OH"^-])/(["H"_2"O"])`
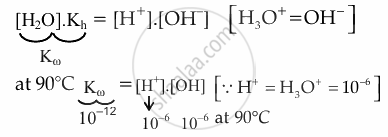
Kω = 10−12
shaalaa.com
Is there an error in this question or solution?
