Advertisements
Advertisements
Question
Answer the following question.
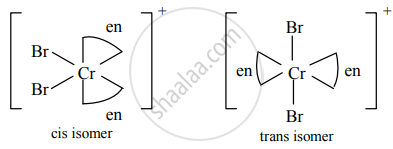
Draw isomers of the following
[Cr(en2)Br2]⊕
Advertisements
Solution
Cis and trans isomers of [Cr(en2)Br2]⊕
a. Cis and trans isomers of [Cr(en2)Br2]+
b. Optical isomers of [Cr(en2)Br2]+

APPEARS IN
RELATED QUESTIONS
Write the structural formula and IUPAC names of all possible isomers of the compound with molecular formula C3H8O.
Answer the following in one or two sentences.
Predict whether the [Cr(en)2(H2O)2]3+ complex is chiral. Write the structure of its enantiomer.
Answer the following question.
Draw geometric isomers and enantiomers of the following complex.
[Pt(en)2ClBr]2⊕
The pair [Co(NH3)5(SO4)]Br and [Co(NH3)5Br]SO4 exhibits ____________ isomerism
Write the type of isomerism exhibited by [Co(NH3)5(NO2)]2+ and [Co(NH3)5ONO]2+ pair of complex ion.
Define the term Hydrated isomers.
Which one of the following pairs represents linkage isomers?
Draw all possible geometrical isomers of the complex \[\ce{[Co(en)2Cl2]^+}\] and identify the optically active isomer.
____________ isomers are formed when the ligand has two different donor atoms.
How many donor groups are present in diethylene triamine?
What type of isomerism is present between (I) [Cr(H2O)6]Cl3 and (II) [Cr(H2O)5Cl]Cl2.H2O?
Indicate the complex ion which shows geometrical isomerism.
The relationship between compound (i) and (ii) is
 |
 |
| (i) | (ii) |
Which of the following has an optical isomer?
Which of the following shows maximum number of isomers?
Which of the following compound is optically active?
Which among the following solid is a non-polar solid?
Give cis isomer of [Co(NH3)4Cl2]⊕.
Give trans isomer of [Co(NH3)4Cl2]⊕.
Indicate the type of isomerism exhibited by the following complex and draw the structures for this isomer:
\[\ce{[Pt(NH3)(H2O)Cl2]}\]
Indicate the types of isomerism exhibited by the following complexes and draw the structures for these isomers: [Pt(NH3)(H2O)Cl2]
Write structures for geometrical isomers of diamminebromochloroplatinum (II).
Give a chemical test to show that \[\ce{[Co(NH3)5Cl]SO4}\] and \[\ce{[Co(NH3)5SO4]CI}\] are ionisation isomers.
Which one of the following complex ions has geometrical isomers?
