Advertisements
Advertisements
Question
Answer the following in brief.
Calculate the packing efficiency of metal crystal that has simple cubic structure.
Advertisements
Solution
Packing efficiency of metal crystal in the simple cubic lattice:
- Step 1: Radius of sphere:
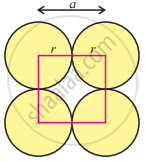
In the simple cubic unit cell, particles (spheres) are at the corners and touch each other along the edge.
A face of a simple cubic unit cell is shown in the figure.
From the figure, we can find that
a = 2r or r = `"a"/2` ......(1)
where, ‘r’ is the radius of atom and ‘a’ is the length of the unit cell edge. - Step 2: Volume of sphere:
Volume of a sphere = `4/3π"r"^3`.
Substitution for r from equation (1) gives:
Volume of one particle = `4/3π ("a"/2)^3` = `(pi"a"^3)/6` ....(2) - Step 3: Total volume of particles:
Because a simple cubic unit cell contains only one particle, the volume occupied by the particle in unit cell =`(pi"a"^3)/6` - Step 4: Packing efficiency:
Packing efficiency = `"Volume occupied by particles in unit cell"/"Total volume of a unit cell" xx 100`
`= (pi"a"^3//6)/"a"^3 xx 100 = (100 pi)/6 = (100 xx 3.142)/6 = 52.36 %`
Thus, in a simple cubic lattice, 52.36 % of total space is occupied by particles and 47.64% is empty space, that is, void volume.
APPEARS IN
RELATED QUESTIONS
Answer the following in one or two sentences.
Mention two properties that are common to both hcp and ccp lattices.
Answer the following in brief.
Cesium chloride crystallizes in a cubic unit cell with Cl– ions at the corners and a Cs+ ion in the center of the cube. How many CsCl molecules are there in the unit cell?
Cu crystallizes in fcc unit cell with edge length of 495 pm. What is the radius of Cu atom?
The density of iridium is 22.4 g/cm3. The unit cell of iridium is fcc. Calculate the radius of iridium atom. Molar mass of iridium is 192.2 g/mol.
Aluminium crystallizes in a cubic close-packed structure with a unit cell edge length of 353.6 pm. What is the radius of Al atom? How many unit cells are there in 1.00 cm3 of Al?
Calculate the packing efficiency for bcc lattice.
A compound of X and Y crystallizes in ccp structure in which the X atoms occupy the lattice points at the corners of cube and Y atoms occupy the centres of each of the cube faces. The formula of this compound is ____________.
The total volume occupied by particles m simple cubic unit cell is ____________.
The number of particles in 1 g of a metallic crystal is equal to ____________.
The coordination number of each sphere in simple cubic lattice is ____________.
Which of the following contains the highest number of atoms?
Which among the following crystal structures the edge length of unit cell is equal to twice the radius of one atom?
What is the edge length of fcc type of unit cell having density and atomic mass 6.22 g cm−3 and 60 g respectively?
1 mol of CO2 contains ____________.
Which of the following is INCORRECT?
Atoms of elements A and B crystallize in hep lattice to form a molecule. Element A occupies 2/3 of tetrahedral voids, the formula of molecule is ______.
Copper crystallises with fee unit cell. If the radius of copper atom is 127.8 pm, calculate the density of copper? (At. mass: Cu = 63.55 g mol-1)
What is the percentage of void space in bcc type of in unit cell?
An element crystallizes in a bee lattice with cell edge of 500 pm. The density of the element is 7.5 g cm-3. How many atoms are present in 300 g of metal?
Silver crystallizes in face centred cubic structure, if radius of silver atom is 144.5 pm. What is the edge length of unit cell?
Gold crystallizes in face centred cubic structure. If atomic mass of gold is 197 g mol-1, the mass of unit cell of gold is ______.
A compound made of elements C and D crystallizes in a fee structure. Atoms of C are present at the corners of the cube. Atoms of D are at the centres of the faces of the cube. What is the formula of the compound?
The packing efficiency of bcc is ______.
