Advertisements
Advertisements
Question
An element Y has a valency of 4. Write the formula for its :
- chloride
- oxide
- sulphate
- carbonate
- nitrate
Advertisements
Solution
Formula for the listed compounds are as follows:
- Crossing the valencies, chloride for the element Y will have the chemical formula YCl4.

- Crossing over the valencies, oxide for the element Y will have the chemical formula YO2.
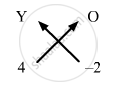
- Crossing the valencies, sulphate for the element Y will have the chemical formula Y(SO4)2.

- Crossing the valencies, carbonate for the element Y will have the chemical formula Y(CO3)2.

- Crossing the valencies, nitrate for the element Y will have the chemical formula Y(NO3)4.
APPEARS IN
RELATED QUESTIONS
Write down the formula of aluminium chloride.
What is meant by the symbol of an element ? Explain with examples.
An element X has a valency of 4 whereas another element Y has a valency of 1. What will be the formula of the compound formed between X and Y ?
Correct the following statement:
Molecular formula of water is H2O2
Name the element in the compound and give the formula of the following compound:
Carbonic acid
Write the formula of the following compound:
Aluminium sulphate
Write the formula of the following compound:
Iron [II] oxide
Write the formula of the following compound:
Iron [III] oxide
Write the formula of the following compound:
Lead [II] chloride
There is ______ oxygen atom is water molecule.
