Advertisements
Advertisements
Question
Account for the following.
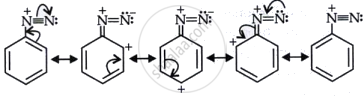
Diazonium salts of aromatic amines are more stable than those of aliphatic amines.
Short Answer
Advertisements
Solution
Diazonium salts of aromatic amines are stabilized by resonance with the aromatic ring, allowing the positive charge on the nitrogen to be delocalized into the ring. The delocalization enhances the stability of the diazonium ion. Conversely, aliphatic amines lack this resonance stabilization, resulting in their diazonium salts being less stable and more susceptible to breakdown. Resonance in diazonium salt, as shown below.
shaalaa.com
Is there an error in this question or solution?
