Advertisements
Advertisements
Question
According to Stefan’s law of radiation, a black body radiates energy σT4 from its unit surface area every second where T is the surface temperature of the black body and σ = 5.67 × 10–8 W/m2K4 is known as Stefan’s constant. A nuclear weapon may be thought of as a ball of radius 0.5 m. When detonated, it reaches temperature of 106 K and can be treated as a black body.
- Estimate the power it radiates.
- If surrounding has water at 30°C, how much water can 10% of the energy produced evaporate in 1s? [Sw = 4186.0 J/kg K and Lv = 22.6 × 105 J/kg]
- If all this energy U is in the form of radiation, corresponding momentum is p = U/c. How much momentum per unit time does it impart on unit area at a distance of 1 km?
Advertisements
Solution
Given, σ = 5.67 × 10–8 W/m2 kg
Radius = R = 0.5 m, T = 106 K
a. Power radiated by Stefan's law
P = σAT4 = (4πR2)T4
= 5.67 × 10–4 × 4 × (3.14) × (0.5)2 × (106)4
= 1.78 × 1017 J/s
= 1.8 × 1017 J/s
b. Energy available per second, U = 1.8 × 1017 J/s = 18 × 1016 J/s
Actual energy required to evaporate water = 10% of 1.8 × 1017 J/s
= 1.8 × 1016 J/s
Energy used per second to raise the temperature of m kg of water 30°C to 100°C and then into vapour at 100°C
= msw Δθ + mLv
= m × 4186 × (100 – 30) + m × 22.6 × 105
= 2.93 × 105 m + 22.6 × 105
m = 25.53 × 105 m J/s
As per the question, 25.53 × 105 m = 1.8 × 1016
or m = `(1.8 xx 10^16)/(25.33 xx 10^5) = 7.0 xx 10^9` kg
c. Momentum per unit time,
p = `U/c = U/c`
= `(1.8 xx 10^17)/(3 xx 10^8)`
= `6 xx 10^8` kg-m/s2 ......`[(P = "momentum"),(V = "energy"),(C = "velocity of light")]`
Momentum per unit time per unit
Area p = `P/(4piR^2)`
= `(6 xx 10^8)/(4 xx 3.14 xx (10^3)^2`
⇒ d = 47.7 N/m2 .......[4πR2 = Surface area]
APPEARS IN
RELATED QUESTIONS
Find the ratio of the lengths of an iron rod and an aluminium rod for which the difference in the lengths is independent of temperature. Coefficients of linear expansion of iron and aluminium are 12 × 10–6 °C–1 and 23 × 10–6 °C–1 respectively.
An aluminium plate fixed in a horizontal position has a hole of diameter 2.000 cm. A steel sphere of diameter 2.005 cm rests on this hole. All the lengths refer to a temperature of 10 °C. The temperature of the entire system is slowly increased. At what temperature will the ball fall down? Coefficient of linear expansion of aluminium is 23 × 10–6 °C–1 and that of steel is 11 × 10–6 °C–1.
A glass window is to be fit in an aluminium frame. The temperature on the working day is 40°C and the glass window measures exactly 20 cm × 30 cm. What should be the size of the aluminium frame so that there is no stress on the glass in winter even if the temperature drops to 0°C? Coefficients of linear expansion for glass and aluminium are 9.0 × 10–6 °C–1 and 24 ×100–6°C–1 , respectively.
One end of a steel rod (K = 46 J s−1 m−1°C−1) of length 1.0 m is kept in ice at 0°C and the other end is kept in boiling water at 100°C. The area of cross section of the rod is 0.04 cm2. Assuming no heat loss to the atmosphere, find the mass of the ice melting per second. Latent heat of fusion of ice = 3.36 × 105 J kg−1.
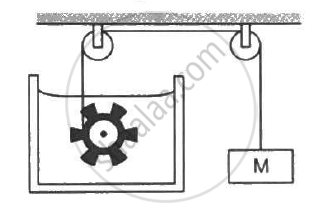
Following Figure shows water in a container having 2.0 mm thick walls made of a material of thermal conductivity 0.50 W m−1°C−1. The container is kept in a melting-ice bath at 0°C. The total surface area in contact with water is 0.05 m2. A wheel is clamped inside the water and is coupled to a block of mass M as shown in the figure. As the block goes down, the wheel rotates. It is found that after some time a steady state is reached in which the block goes down with a constant speed of 10 cm s−1 and the temperature of the water remains constant at 1.0°C. Find the mass M of the block. Assume that the heat flows out of the water only through the walls in contact. Take g = 10 m s−2.
On a winter day when the atmospheric temperature drops to −10°C, ice forms on the surface of a lake. (a) Calculate the rate of increase of thickness of the ice when 10 cm of the ice is already formed. (b) Calculate the total time taken in forming 10 cm of ice. Assume that the temperature of the entire water reaches 0°C before the ice starts forming. Density of water = 1000 kg m−3, latent heat of fusion of ice = 3.36 × 105 J kg−1and thermal conductivity of ice = 1.7 W m−1°C−1. Neglect the expansion of water of freezing.
A semicircular rod is joined at its end to a straight rod of the same material and the same cross-sectional area. The straight rod forms a diameter of the other rod. The junctions are maintained at different temperatures. Find the ratio of the heat transferred through a cross section of the semicircular rod to the heat transferred through a cross section of the straight rod in a given time.
Following figure shows two adiabatic vessels, each containing a mass m of water at different temperatures. The ends of a metal rod of length L, area of cross section A and thermal conductivity K, are inserted in the water as shown in the figure. Find the time taken for the difference between the temperatures in the vessels to become half of the original value. The specific heat capacity of water is s. Neglect the heat capacity of the rod and the container and any loss of heat to the atmosphere.

A calorimeter of negligible heat capacity contains 100 cc of water at 40°C. The water cools to 35°C in 5 minutes. The water is now replaced by K-oil of equal volume at 40°C. Find the time taken for the temperature to become 35°C under similar conditions. Specific heat capacities of water and K-oil are 4200 J kg−1 K−1 and 2100 J kg−1 K−1respectively. Density of K-oil = 800 kg m−3.
There are two identical vessels filled with equal amounts of ice. The vessels are of different metals. If the ice melts in the two vessels in 20 and 35 minutes respectively, the ratio of the coefficients of thermal conductivity of the two metals is:
The coefficient of thermal conductivity depends upon ______.
Heat is associated with ______.
These days people use steel utensils with copper bottom. This is supposed to be good for uniform heating of food. Explain this effect using the fact that copper is the better conductor.
A cylinder of radius R made of material of thermal conductivity K1 is surrounded by a cylindrical shell of inner radius R and outer radius 3R made of a material of thermal conductivity K2. The two ends of the combined system are maintained at two different temperatures. What is the effective thermal conductivity of the system?
In conduction, how does heat mainly travel through a solid rod kept with one end in a flame?
Which of the following is a correct example of a bad conductor of heat used in daily life to reduce heat loss or gain?
During conduction along a metal rod, which part becomes hot first, and why?
