Advertisements
Advertisements
Question
A relevant portion of β-chain of haemoglobin of a normal human is given below:
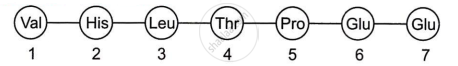
The codon for the sixth amino acid is GAG. The sixth codon GAG mutates to CAA as a result of mutation ‘A’ and into GUG as a result of mutation ‘B’. Haemoglobin structure did not change as a result of mutation ‘A’ whereas haemoglobin structure changed because of mutation ‘B’ leading to sickle-shaped RBCs. Explain giving reasons how could mutation ‘B’ change the haemoglobin structure and not mutation ‘A’.
Advertisements
Solution
The alteration in haemoglobin structure caused by mutation 'B' results from the substitution of glutamic acid with valine at the sixth position of the β-globin chain. This amino acid change originates from a single nucleotide substitution in the β-globin gene, where the codon GAG is replaced by GUG. Under conditions of low oxygen, the mutated haemoglobin molecules polymerize, leading to a deformation in red blood cells. Consequently, the red blood cells change their shape from the normal biconcave discs into elongated, sickle-like forms characteristic of sickle-cell anaemia.
