Advertisements
Advertisements
Question
A gas is initially at a pressure of 100 kPa and its volume is 2.0 m3. Its pressure is kept constant and the volume is changed from 2.0 m3 to 2.5 m3. Its Volume is now kept constant and the pressure is increased from 100 kPa to 200 kPa. The gas is brought back to its initial state, the pressure varying linearly with its volume. (a) Whether the heat is supplied to or extracted from the gas in the complete cycle? (b) How much heat was supplied or extracted?
Advertisements
Solution
(a) Given:-
P1 = 100 kPa,
V1 = 2 m3
V2 = 2.5 m3
∆V = 0.5 m3

Work done, W = P∆V
\[W = 100 \times {10}^3 \times 0 . 5\]
\[W = 5 \times {10}^4 J\]
WAB = Area under line AB = 5 × 104 J
If volume is kept constant for line BC, then ∆V = 0.
WBC = P∆V = 0
Work done while going from point B to C = 0
When the system comes back to the initial point A from C, work done is equal to area under line AC.
WCA = Area of triangle ABC + Area of rectangle under line AB
Total work done, W = Area enclosed by the ABCA
W = WAC - WAB
From the graph, we see that the area under AC is greater than the area under AB. We also see that heat is extracted from the system as change in the internal energy is zero.
(b) Amount of heat extracted = Area enclosed under ABCA
\[= \frac{1}{2} \times 0 . 5 \times 100 \times {10}^3 = 25000 J\]
APPEARS IN
RELATED QUESTIONS
Explain why Two bodies at different temperatures T1 and T2, if brought in thermal contact, do not necessarily settle to the mean temperature (T1 + T2)/2.
Explain why Air pressure in a car tyre increases during driving.
Two cylinders A and B of equal capacity are connected to each other via a stopcock. A contains a gas at standard temperature and pressure. B is completely evacuated. The entire system is thermally insulated. The stopcock is suddenly opened. Answer the following:
Do the intermediate states of the system (before settling to the final equilibrium state) lie on its P-V-T surface?
A closed bottle contains some liquid. the bottle is shaken vigorously for 5 minutes. It is found that the temperature of the liquid is increased. Is heat transferred to the liquid? Is work done on the liquid? Neglect expansion on heating.
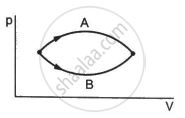
Figure shows two processes A and B on a system. Let ∆Q1 and ∆Q2 be the heat given to the system in processes A and B respectively. Then ____________ .
A gas is contained in a metallic cylinder fitted with a piston. The piston is suddenly moved in to compress the gas and is maintained at this position. As time passes the pressure of the gas in the cylinder ______________ .
A gas is taken along the path AB as shown in figure. If 70 cal of heat is extracted from the gas in the process, calculate the change in the internal energy of the system.

Which of the following system freely allows the exchange of energy and matter with its environment?
Define heat.
A system releases 100 kJ of heat while 80 kJ of work is done on the system. Calculate the change in internal energy.
Explain given cases related to energy transfer between the system and surrounding –
- energy transferred (Q) > 0
- energy transferred (Q) < 0
- energy transferred (Q) = 0
8 m3 of a gas is heated at the pressure 105 N/m2 until its volume increases by 10%. Then, the external work done by the gas is ____________.
Two samples A and B, of a gas at the same initial temperature and pressure are compressed from volume V to V/2; A isothermally and B adiabatically. The final pressure of A will be ______.
Two cylinders A and B of equal capacity are connected to each other via a stopcock. A contains a gas at standard temperature and pressure. B is completely evacuated. The entire system is thermally insulated. The stopcock is suddenly opened. Answer the following:
What is the change in the temperature of the gas?
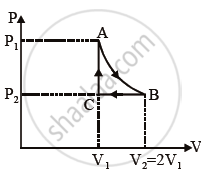
n mole of a perfect gas undergoes a cyclic process ABCA (see figure) consisting of the following processes:
A `→` B: Isothermal expansion at temperature T so that the volume is doubled from V1 to V2 = 2V1 and pressure changes from P1 to P2.
B `→` C: Isobaric compression at pressure P2 to initial volume V1.
C `→` A: Isochoric change leading to change of pressure from P2 to P1.
Total workdone in the complete cycle ABCA is ______.
In thermodynamics, heat and work are ______.
What is heat?
A system releases 125 kJ of heat while 104 kJ work is done on the system. Calculate the change in internal energy.
Explain the change in internal energy of a thermodynamic system (the gas) by heating it.
