Advertisements
Advertisements
Question
3, 3-dimethyl butane-2-ol on treatment with conc. H2SO4 to give tetramethyl ethylene as a major product. Suggest a suitable mechanism.
Advertisements
Solution
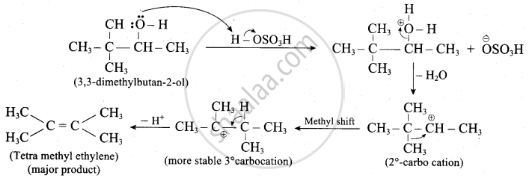
According to Saytzeff’s rule the dehydration of 3, 3-dimethyl butane-2-ol gives a mixture of alkenes. But the secondary carbocation formed in this reaction undergoes rearrangement to form a more stable tertiary carbocation which further, undergoes 13 elimination leads more stable product, that is 2, 3-dimethyl but-2-ene (more yield). According to Saytzeff’s rule, 2, 3-dimethyl pent-2-ene is the major product.
APPEARS IN
RELATED QUESTIONS
Carbolic acid is ____________.
Which one of the following will react with phenol to give salicylaldehyde after hydrolysis.
Complete the following reaction.
Draw the major product formed when 1-ethoxyprop-1-ene is heated with one equivalent of HI
What will be the product (X and A)for the following reaction
acetylchloride \[\ce{{acetylchloride}->[i)CH3MgBr][ii)H3O+]X ->[acidK2Cr2O7] A}\]
Draw the major product formed when 1-ethoxyprop-1-ene is heated with one equivalent of HI.
Predict the major product, when 2-methyl but -2-ene is converted into an alcohol in the following method.
Acid catalysed hydration
Draw the major product formed when 1-ethoxyprop-1-ene is heated with one equivalent of HI.
Draw the major product formed when 1-ethoxyprop-1-ene is heated with one equivalent of HI.
The correct IUPAC name of the compound,
\[\begin{array}{cc}
\ce{CH3}\phantom{......}\\
|\phantom{........}\\
\ce{H3C - CH - CH - CH - CH2 - OH}\\
|\phantom{............}|\phantom{........}\\
\ce{Cl}\phantom{...........}\ce{CH3}\phantom{......}
\end{array}\]
