Please select a subject first
Advertisements
Advertisements
Define the degree of dissociation.
Concept: undefined >> undefined
Answer the following :
Derive the relationship between the degree of dissociation and dissociation constant in weak electrolytes.
Concept: undefined >> undefined
Advertisements
Choose the most correct option.
The order of the reaction for which the units of the rate constant are mol dm-3 s-1 is _______.
Concept: undefined >> undefined
Choose the most correct option.
The rate constant for the reaction \[\ce{2N2O5_{(g)} -> 2N2O4_{(g)} + O2_{(g)}}\] is `4.98 xx 10^-4 "s"^-1`. The order of reaction is _____________.
Concept: undefined >> undefined
The time required for 90% completion of a certain first-order reaction is t. The time required for 99.9% completion will be _________.
Concept: undefined >> undefined
Choose the most correct option.
Slope of the graph ln[A]t versus t for first-order reaction is _________.
Concept: undefined >> undefined
What is the half life of a first order reaction if time required to decrease concentration of reactant from 0.8 M to 0.2 M is 12 h?
Concept: undefined >> undefined
Choose the most correct option.
The reaction, \[\ce{3ClO- -> ClO^-3 + 2Cl-}\] occurs in two steps,
(i) \[\ce{2ClO- -> ClO^-2}\]
(ii) \[\ce{ClO^-2 + ClO- -> ClO^-_3 + Cl-}\]
The reaction intermediate is _______.
Concept: undefined >> undefined
The elementary reaction \[\ce{O3_{(g)} + O_{(g)} -> 2O2_{(g)}}\] is ______.
Concept: undefined >> undefined
Choose the most correct option.
Rate law for the reaction, \[\ce{2NO + Cl2 -> 2NOCl}\] is rate = k[NO2]2[Cl2]. Thus of k would increase with _____________.
Concept: undefined >> undefined
Choose the most correct option.
For an endothermic reaction, X ⇌ Y. If Ef is the activation energy of the forward reaction and Er that for the reverse reaction, which of the following is correct?
Concept: undefined >> undefined
Answer the following in one or two sentences.
For the reaction, \[\ce{CH3Br_{(aq)} + OH^{-}_{(aq)} -> CH3OH^{\ominus}_{(aq)} + Br^{\ominus}_{(aq)}}\], rate law is rate = \[\ce{k[CH3Br][OH^\ominus]}\]
How does reaction rate changes if \[\ce{[OH^\ominus]}\] is decreased by a factor of 5?
Concept: undefined >> undefined
Answer the following in brief.
For the reaction 2A + B → products, find the rate law from the following data.
| [A]/M | [B]/M | rate/M s-1 |
| 0.3 | 0.05 | 0.15 |
| 0.6 | 0.05 | 0.30 |
| 0.6 | 0.2 | 1.20 |
Concept: undefined >> undefined
Answer in brief.
What are bidentate ligands? Give one example.
Concept: undefined >> undefined
Answer in brief.
What are the coordination number and oxidation state of the metal ion in the complex [Pt(NH3)Cl5]-?
Concept: undefined >> undefined
Answer in brief.
What is the difference between a double salt and a complex? Give an example.
Concept: undefined >> undefined
Write IUPAC name of the following compound.
\[\begin{array}{cc}
\ce{CH3 - CH = C - CH - Br}\\
\phantom{......}|\phantom{....}|\\
\ce{\phantom{......}H3C} \ce{\phantom{...}CH3}
\end{array}\]
Concept: undefined >> undefined
Write IUPAC name of the following compound.
\[\begin{array}{cc}
\ce{CH3 - CH - CH - CH2 - CH3}\\
|\phantom{......}|\phantom{.........}\\
\ce{Cl} \ce{\phantom{....}CH3\phantom{.......}}
\end{array}\]
Concept: undefined >> undefined
Write IUPAC name of the following compound.
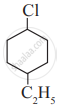
Concept: undefined >> undefined
Write the IUPAC name of the following compound.
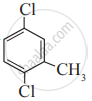
Concept: undefined >> undefined
